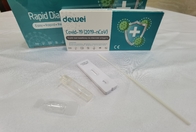
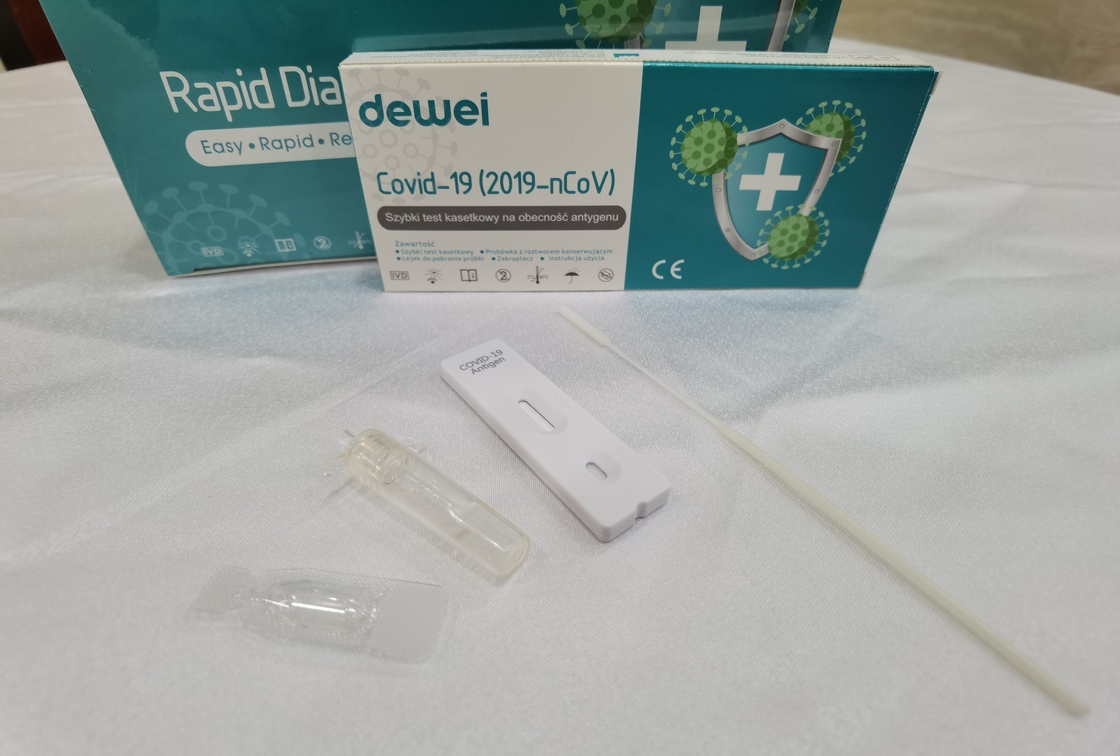
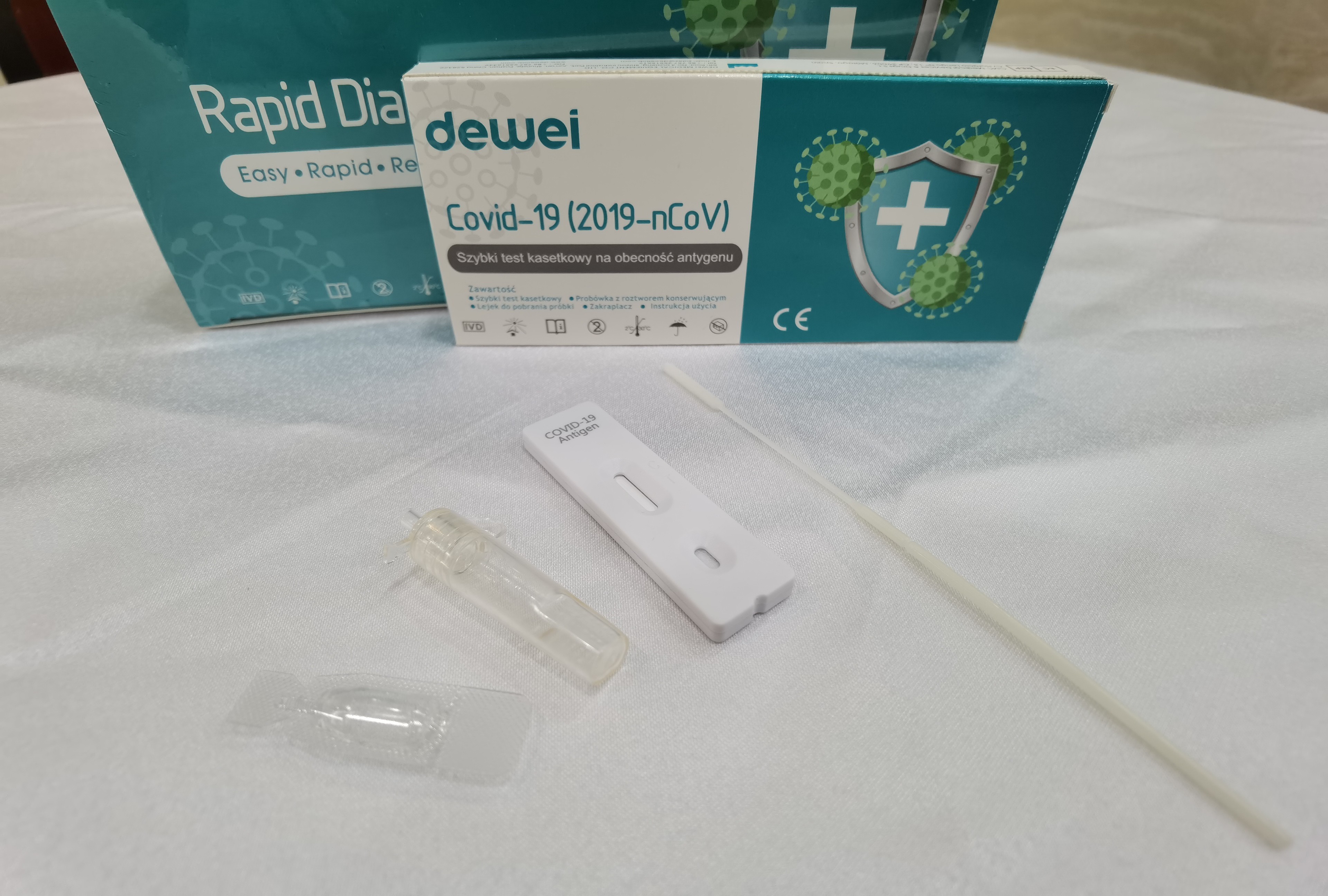
Fast Check Nasal Nasopharyngeal Swab 15mins Reading Antigen Sample Corona Rapid Test Cassette
Colloidal Gold Assay
² Easy: One Step
² Fast: 15 Minutes
² Cheap: Lower cost with high efficiency
² Convenient: Throat/Nasal swab sample
INTENDED USE OF ANTIGEN RAPID TEST
One Step 2019-Novel Coronavirus (2019-nCoV) Antigen Test Cassette is an in vitro diagnostic test device intended for the rapid detection of 2019- nCoV antigen in nasopharyngeal and oropharyngeal swab from patients with suspected 2019-nCoV infection, patients with suspected clustering cases, and others who need to diagnose or differentially diagnose 2019-Novel Coronavirus (2019-nCoV).
MAIN CONTENTS OF ANTIGEN RAPID TEST
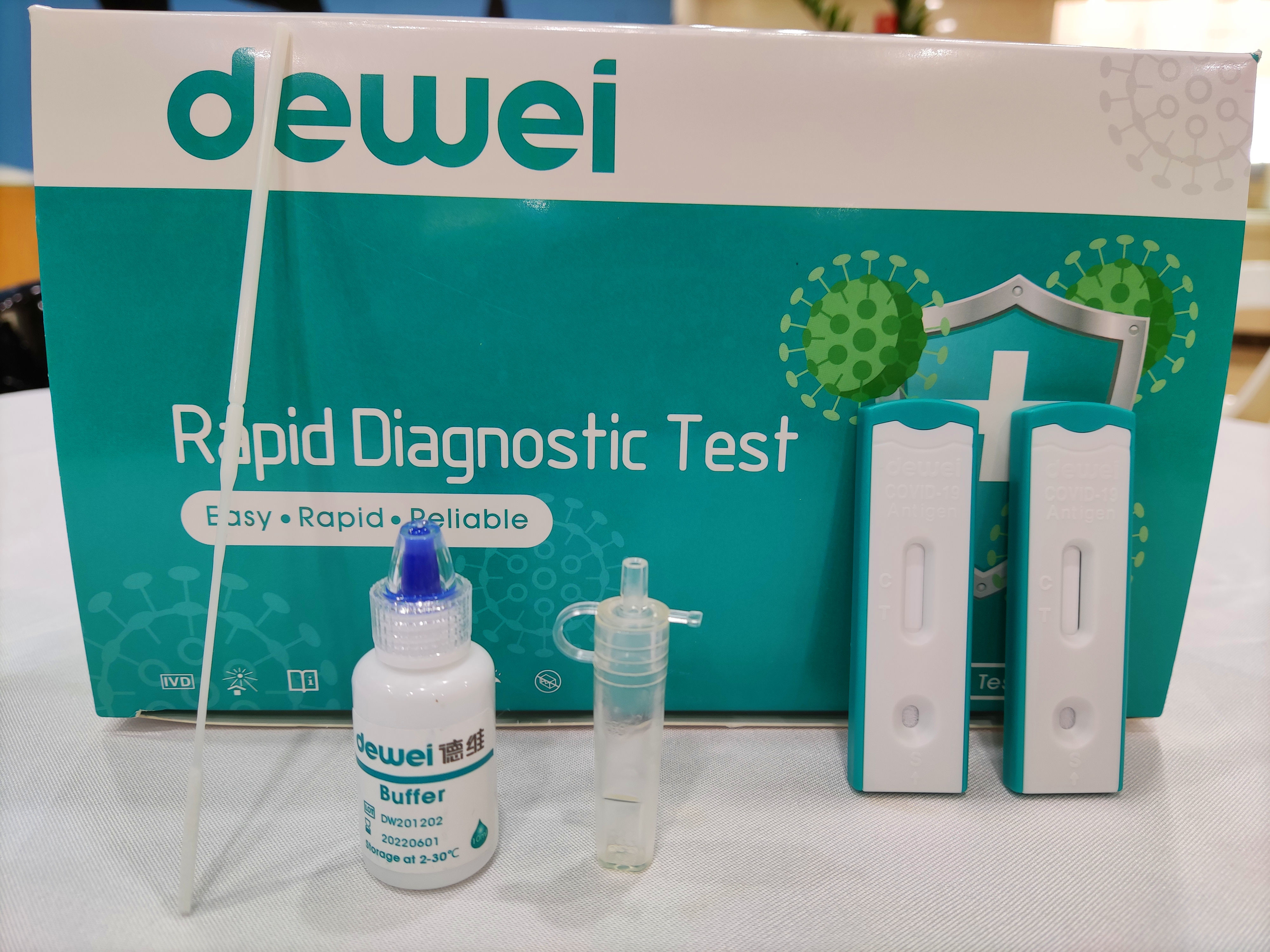
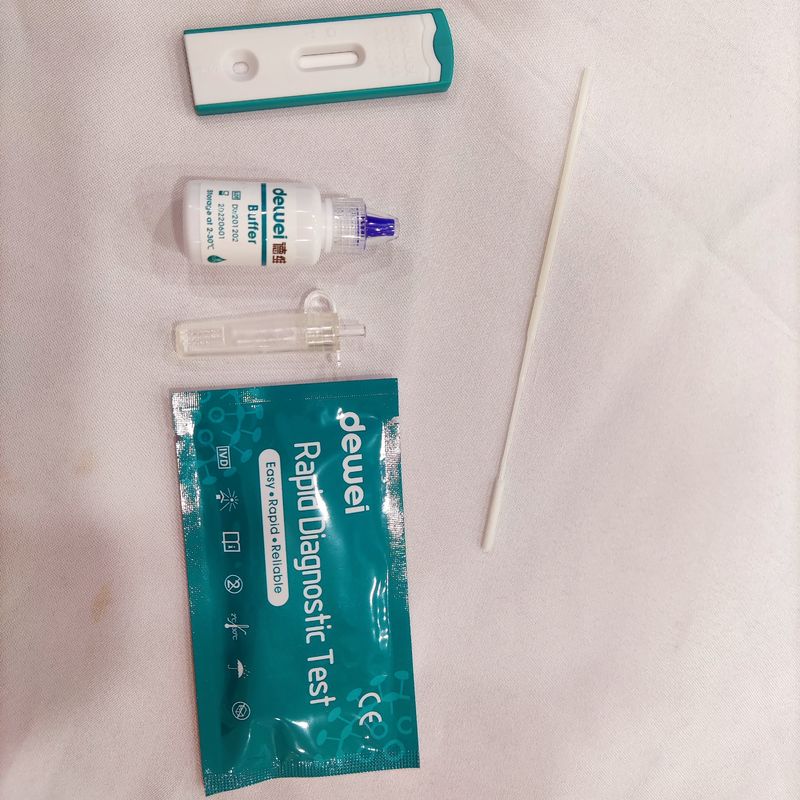
• Rapid Test Cassette with desiccant;
• Extraction Tube;
• Extraction Reagent;
• Instructions for use;
• Flocked Swab;
WHAT ELSE DO YOU NEED ?
1.Sterilized nasopharyngeal swabs for sample collection or sterilized oropharyngeal swabs for sample collection
2. Medical mask and medical latex gloves
3. Watch or timer
PRECAUTIONS
• For in vitro diagnostic use only.
• Do not use after expiration date.
• The test Cassette should remain in the sealed pouch until use.
• The used test Cassette should be discarded according to local regulations.
STORAGE AND STABILITY
• Store at 39 ~ 86 º F (4 ~ 30 º C) in the sealed pouch up to the expiration date.
• Keep away from direct sunlight, moisture and heat.
• DO NOT FREEZE.
SPECIMEN COLLECTION AND PREPARATION
1.Specimen to be tested should be obtained and handled by standard methods for their collections.
2. Nasopharyngeal swab specimen:
To collect nasopharyngeal specimen pharyngeal wall surely so as to rub a part near lower respiratory tract, carefully insert the sterile swab into the nostril that presents the most secretion under visual inspection. Using gentle rotation, push the swab til resistance is met at the level of the turbinates (less than one inch into the nostril). Rotate the swab several times against the nasal wall.
3. Oropharyngeal swab specimen:
Insert swab from oral cavity into pharynx slowly and collect mucous membrane epidermis by rubbing posterior pharyngeal wall or faucial tonsil several times. Antigen of enough quantity cannot be collected with upper respiratory tract. Collection specimen by letting the spherical trip touch the part near posterior. In addition, do not use nasopharyngeal swab when collecting samples as it may cause insufficient collection of specimen.
4. All specimens should be tested as soon as early they are prepared. If necessary, they may be stored at 2-8°C for up to 24 hours or at -20°C for longer periods. Restore the sample to room temperature before test.
HOW TO PERFORM THE TEST?
1. Remove a Test Device from the foil pouch by tearing at the notch and place it on a level surface.
2. Holding Extraction Reagent bottle vertically, add 10 drops (400μL) to the Extraction tube.
3. Insert the nasopharyngeal (and oropharyngeal) swab sample(s) into the extraction solution, then, mix the swab 10 times.
4. Remove the swabs while pressing against the solution tube in order to extract most of the specimen
5. Place the dropper cap and drop 3 drops (60~70μl) into the sample well.
6. Read the result in 10-15 minutes. Do not read results after more than 20 minutes.
Something you need to learn about:
What is a rapid COVID-19 test?
Rapid tests are point-of-care diagnostic tests that use a mucus sample from the nose or throat, but can be analyzed at a doctor’s office or clinic instead of being sent to a laboratory. The results can be available very quickly, sometimes in minutes. In fact, many companies are starting to announce rapid tests that can one day be used at home.
Rapid tests are almost always antigen tests. Traditional diagnostic tests are molecular tests.
What is the difference between an antigen test and a molecular test?
An antigen test detects specific proteins on the surface of the virus. These tests are quicker and less expensive, but have a higher chance of missing an active infection, according to the FDA.
A molecular (PCR or polymerase chain reaction) test detects the virus’s genetic material. These tests require a more complex technology to get results, and it usually takes a day or two to get those results (depending on lab capacity, results may take up to a week).
If an antigen test shows a negative result and you have reason to believe you may have the virus (because of symptoms or exposure), your doctor may order a molecular test to confirm the results.
Which tests are more accurate?
No test is 100% accurate, but the molecular tests are considered to be more accurate than the antigen tests, according to available research.
According to the FDA, an antigen test cannot “definitively rule out active coronavirus infection,” but positive results are “highly accurate” (negative results may require a confirmation test).
Harvard Medical School wrote in August that the reported rate of false negatives with molecular testing is as low as 2% and as high as 37%. A molecular test using a deep nasal swab will have fewer false negative results than samples from throat swabs or saliva, they say.
For antigen testing, Harvard noted that the reported rate of false negative results can be as high as 50%, but that the FDA has granted emergency use authorization for a more accurate antigen test.
In general, some of the issues that may affect the accuracy of a test include ineffective swabbing, contamination or mishandling of the sample, or problems with the testing chemicals, according to the FDA.
If antigen tests are less accurate, why would we use them?
Experts say the value in the rapid antigen tests is in the frequency of the testing rather than the accuracy, with repeated testing recommended. According to Science, getting a false negative in an antigen test two or three times in a row is rare. This strategy lines up with the NPR report mentioned above, which would employ daily testing of those in high-risk areas.
How and where are we currently using rapid tests?
Rapid antigen tests may be used right now some in doctor’s offices and hospitals, but the most visible deployment of the repeated rapid test strategy is by college and professional football teams, who test players and coaches daily to determine if they are healthy for practice and play.
The federal government announced this week it will be sending out 150 million rapid tests to various groups, but distribution to nursing homes so far has been problematic.



 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!