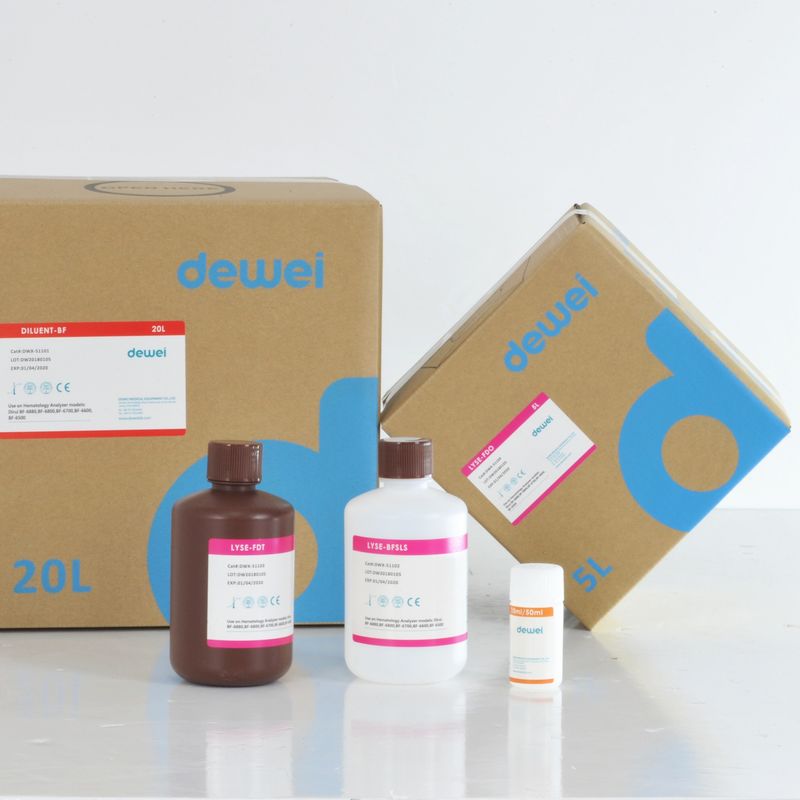
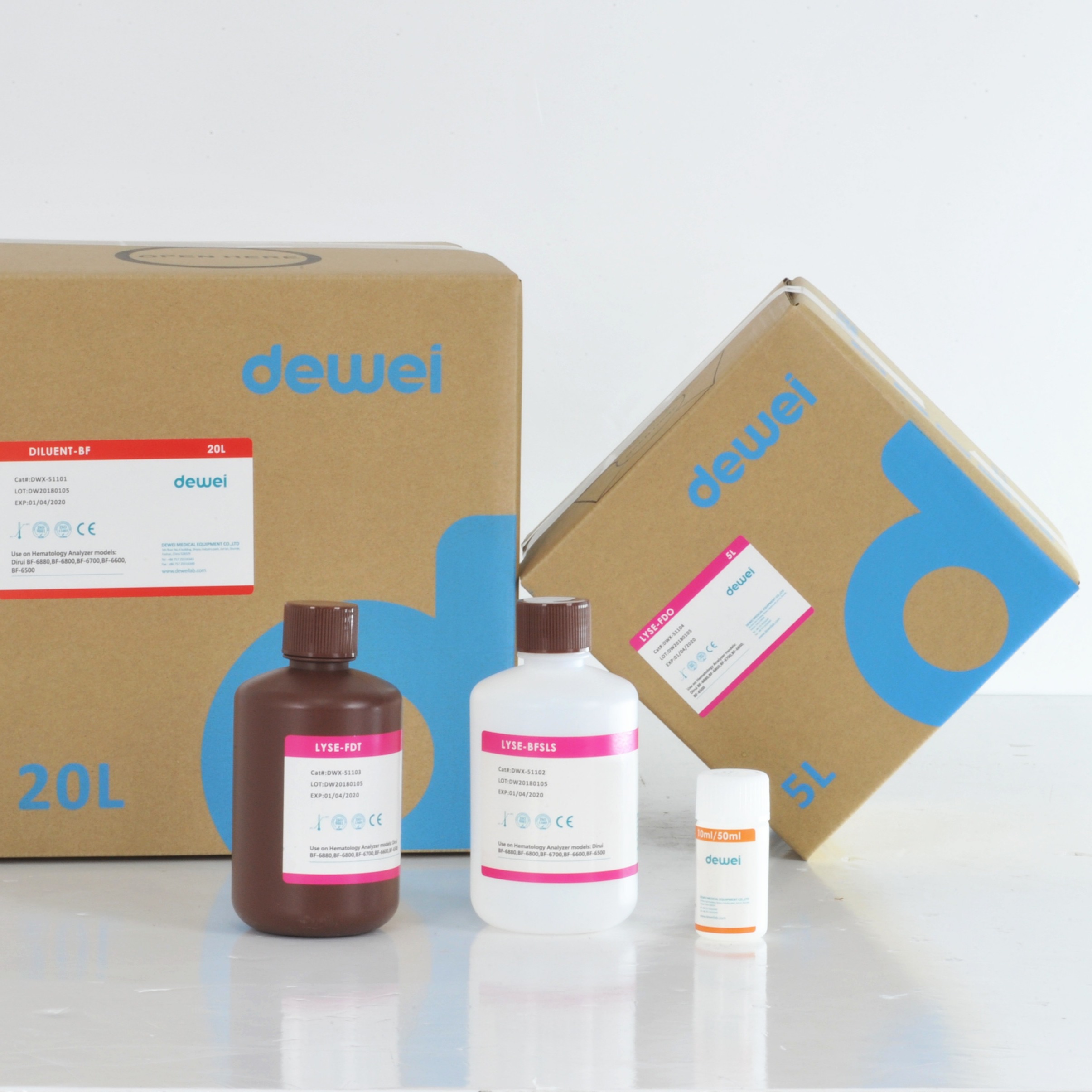
Hematology Analyzer Reagent for DIRUI BF-6700 BF-6600 Diluent Lyse Clean
Specification of Dirui Reagents
| Reagents on Dirui BF-6880 BF-6800 BF-6700 BF-6600 |
| Cat. No |
Description |
Pack Size |
| DWX-51101 |
DILUENT-BF |
20L |
| DWX-51102 |
LYSE-BFSLS |
500ml |
| DWX-51103 |
LYSE-FDT |
500ml |
| DWX-51104 |
LYSE-FDO |
5L |
| DWX-51105 |
RINSE-BF |
10ml/50ml |
Intended Use of Dirui Reagents
To be used for blood cell counting, sizing, and WBC differentiation, hemoglobin determination on DIRUI BF-6700 BF-6600 Hematology Analyzers. This product is for In Vitro Diagnostic Use only, not for home use or self-testing.
PRECAUTIONS
1. Re-calibration is recommended when changing the origin of the reagents.
2. It is not recommended using different brands of solutions together on one unit of analyzer.
3. Do not ingest. Avoid contact with skin and eyes.
4. Skin contact: Flush with plenty of water.
5. Eyes contact: Flush with plenty of water. Obtain immediate medical attention.
6. This kit is for professionals and in vitro diagnostic use only.
7. Never put distilled water on the analyzer (except clean for shipping menu), otherwise the pneumatic unit can be damaged seriously.
STORAGE
1. Store the product at 2-35℃ in a dust free place with good ventilation. Temperature outside the range may damage the product.
2. The open vial life of the product is 60 days. Unused reagents should be discarded after 60 days. Do not mix residual with a new carton.
Frequently Asked Question
1. Can reagents cross use with different manufactures?
Some reagents can cross use but some need to use as a set otherwise the results may be effected. For example, LYSE-4DL, LYSE-4DS,NR-PACK and RET-PACK of SYSMEX XE-2100 model should be used as a set.
2. Compared with other compatible reagent companies, what are our advantages?
It is mainly reflected in the following aspects:
2.1. Professional development of in vitro diagnostic reagents, quality as the first criterion for cooperation.
2.2. Samples and final products are strictly controlled according to international standards.
2.3. The reagent of our company has made a detailed comparison with the original reagent, ensuring that there is no difference with the original reagent, and the quality is stable and reliable.




 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!