【INTENDED USE】
The Strep A Rapid Test (Swab) is a rapid visual immunoassay for the qualitative, presumptive detection of Group A Streptococcus antigens in human throat swab specimens. This kit is intended for use as an aid in the diagnosis of Strep A infection.
【INTRODUCTION】
Beta-hemolytic Group A Streptococcus is a major cause of upper respiratory infections such as tonsillitis, pharyngitis, and scarlet fever. Early diagnosis and treatment of Group A Streptococcal pharyngitis has been shown to reduce the severity of symptoms and further complications, such as rheumatic fever and glomerulonephritis. Conventional methods for detecting Strep A infection are dependent on isolation and subsequent identification of the organism, and often require 24- 48 hours. Recent development of immunological techniques to detect Group A Streptococcal antigen directly from throat swabs allow physicians to diagnose and administer therapy immediately.
【PRINCIPLE】
The Strep A Rapid Test (Swab) detects Group A Streptococcus antigens through visual interpretation of color development on the internal strip. Anti-Strep A antibodies are immobilized on the test region of the membrane. During the test, the specimen reacts with polyclonal anti-Strep A antibodies conjugated to colored particles and precoated onto the sample pad of the test. The mixture then migrates through the membrane by capillary action and interacts with reagents on the membrane. If there is sufficient Strep A antigen in the specimen, a colored band will form at the test region of the membrane. The presence of this colored band indicates a positive result, while its absence indicates a negative result. The appearance of a colored band at the control region serves as a procedural control, indicating that proper volume of specimen has been added and membrane wicking has occurred.
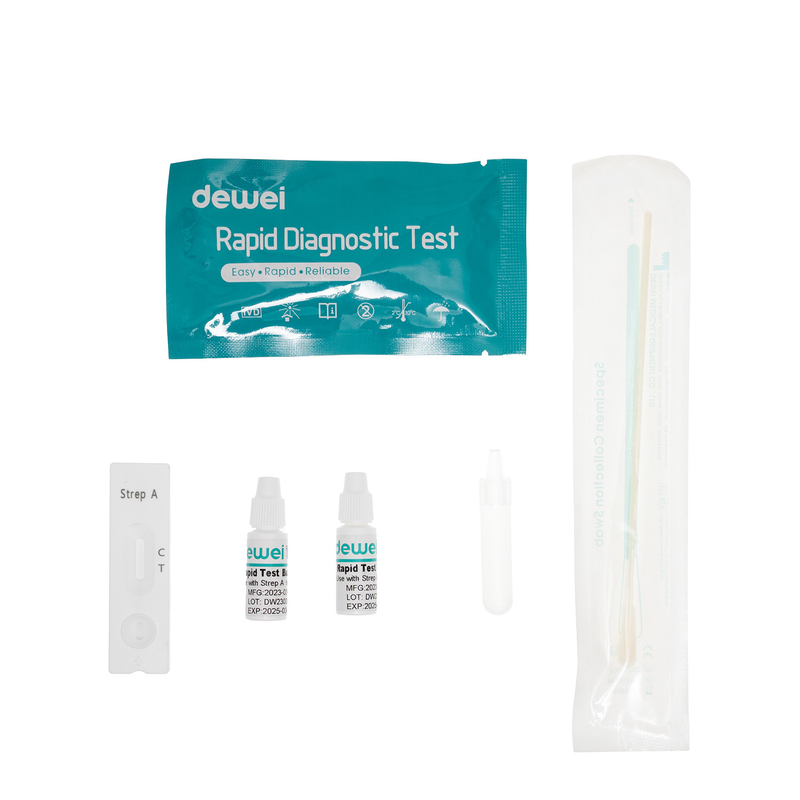
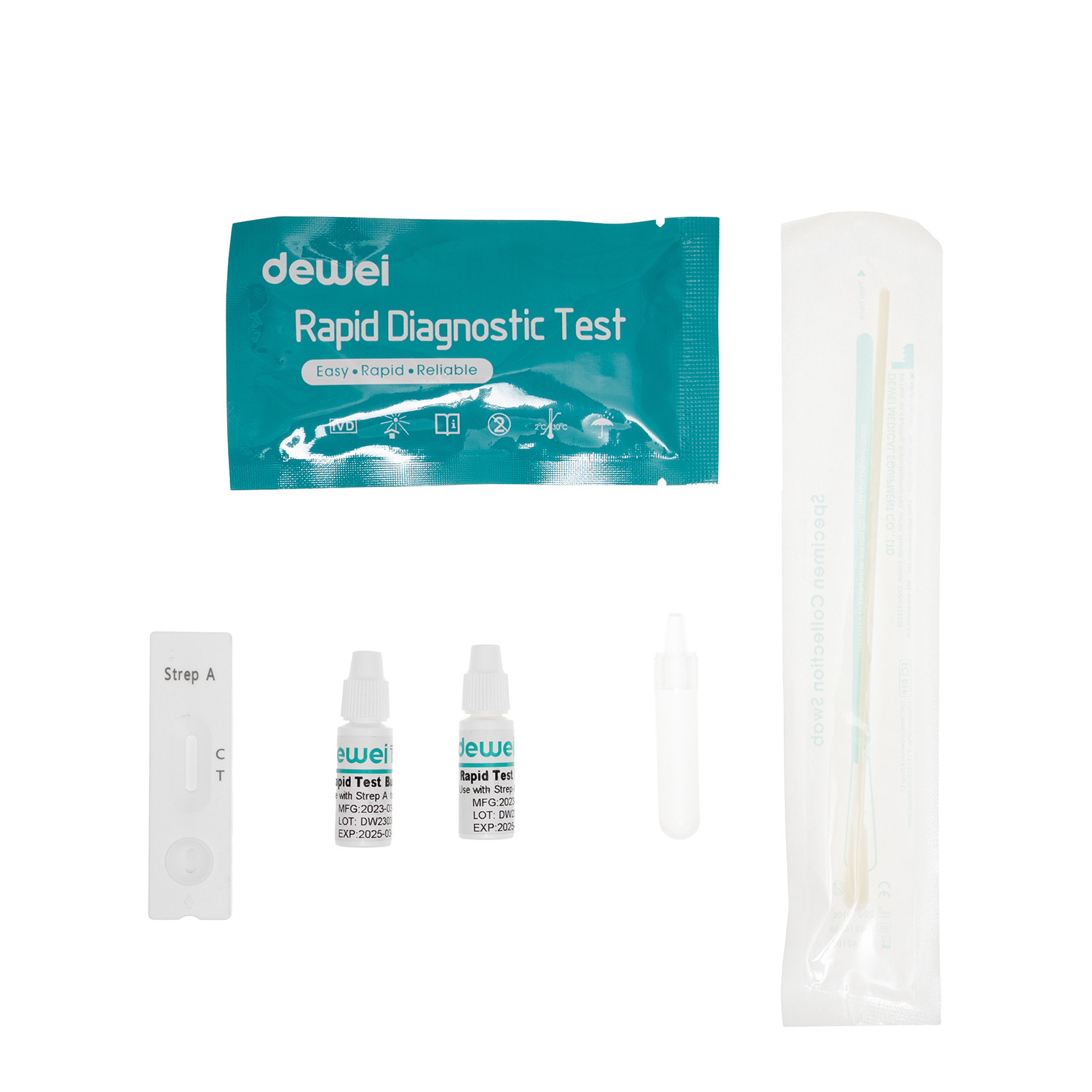
【MAIN CONTENTS】
• Rapid test cassette with desiccant.
• Sampling Swab
• Reagent 1
• Reagent 2
• Extraction Tube
• Package insert
【PRECAUTIONS】
- For professional in vitro diagnostic use only.
- Do not use after the expiration date indicated on the package. Do not use the test if the foil pouch is damaged. Do not reuse tests.
- This kit contains products of animal origin. Certified knowledge of the origin and/or sanitary state of the animals does not completely guarantee the absence of transmissible pathogenic agents. It is therefore recommended that these products be treated as potentially infectious, and handled by observing usual safety precautions (e.g., do not ingest or inhale).
- Avoid cross-contamination of specimens by using a new extraction tube for each specimen obtained.
- Read the entire procedure carefully prior to testing.
- Do not eat, drink or smoke in any area where specimens and kits are handled. Handle all specimens as if they contain infectious agents. Observe established precautions against microbiological hazards throughout the procedure and follow standard procedures for the proper disposal of specimens. Wear protective clothing such as laboratory coats, disposable gloves and eye protection when specimens are assayed.
- Do not interchange or mix reagents from different lots. Do not mix solution bottle caps.
- Use only dacron or rayon tipped sterile swabs with plastic shafts such as those provided. Do not use calcium alginate, cotton tipped, or wooden shafted swabs.
- Reagents 1 & 2 are slightly caustic. Avoid contact with eyes or mucous membranes. In the event of accidental contact, wash thoroughly with water.
- The positive control contain sodium azide, which may react with lead or copper plumbing to form potentially explosive metal azides. When disposing of these solutions always flush with copious amounts of water to prevent azide buildup.
- Humidity and temperature can adversely affect results.
- Used testing materials should be discarded according to local regulations.
【STORAGE AND STABILITY】
• Store at 2 ~ 30 º C in the sealed pouch for 24 months.
• Keep away from direct sunlight, moisture and heat.
• DO NOT FREEZE.
【DIRECTION OF USE】
Bring tests, specimens, buffer and/or controls to room temperature (15- 30°C) before use.
1. Prepare swab specimens:
1) Place a clean extraction tube in the designated area of the workstation. Add 5 drops of Reagent 1 to the extraction tube, then add 4 drops of Reagent 2. Mix the solution by gently swirling the extraction tube.
1) Immediately immerse the swab into the extraction tube. Use a circular motion to roll the swab against the side of the extraction tube so that the liquid is expressed from the swab and can reabsorb.
2) Let stand for 1-2 minutes at room temperature, then squeeze the swab firmly against the tube to expel as much liquid as possible from the swab. Cap the extraction tube with the attached dropper tip. Discard the swab following guidelines for handling infectious agents.
2. Remove the test from its sealed pouch, and place it on a clean, level surface. Label the cassette with patient or control identification. For best results, the assay should be performed within one hour.
3. Add 3 drops (approximately 120 µL) of extracted solution from the extraction tube to the sample well on the test cassette. Avoid trapping air bubbles in the specimen well (S), and do not add any solution to the observation window. As the test begins to work, color will migrate across the membrane.
4. Wait for the colored band(s) to appear. The result should be read at 10 minutes. Do not interpret the result after 15 minutes.

【INTERPRETATION OF RESULTS】
POSITIVE: The presence of two lines as control line (C) and test line (T) within the result window indicates a positive result.
NEGATIVE: The presence of only control line (C) within the result window indicates a negative result.
INVALID: If the control line (C) is not visible within the result window after performing the test, the result is considered invalid. Some causes of invalid results are because of not following the directions correctly or the test may have deteriorated beyond the expiration date. It is recommended that the specimen be re-tested using a new test.
| 1 |
How do you get strep A? |
Strep A infections are spread by close contact with an infected person. They can be passed on through coughs and sneezes or from a wound. In some people, the bacteria live in the body without causing symptoms or making them feel unwell. But they can still pass the bacteria on to others. |
| 2 |
Is strep A usually serious? |
Strep throat is typically a mild condition, but the infection can be very painful. Your sore throat may be severe and very uncomfortable. The lymph nodes in your neck may be very tender and swollen. You may have pain when swallowing. |
| 3 |
What are the symptoms of type A strep? |
Symptoms of group A streptococcal infection
sore, red throat or tonsils.
pus on throat or tonsils.
pain on swallowing.
fever and chills.
swollen and tender lymph nodes (glands) in the neck.
vomiting, nausea and abdominal pain.
headache and muscle aches.
general feeling of unwell (malaise). |
| 4 |
Is strep A the same as strep throat? |
Group A Streptococcus (GAS), also known as Strep A, are bacteria commonly found on the skin or in the throat. Under some circumstances these bacteria can cause disease. GAS infection commonly presents as a mild sore throat ('strep throat') and skin/soft tissue infections such as impetigo and cellulitis. |
| 5 |
Will strep A go away on its own? |
Strep throat typically goes away in three to seven days with or without antibiotic treatment. However, if you don't take antibiotics, you can remain contagious for two to three weeks and are at a higher risk for complications, such as rheumatic fever. |
| 6 |
How long is strep a contagious? |
People who start taking antibiotics to clear up strep throat become less contagious in about 24 hours. However, people who don't take antibiotics can spread the bacteria for a couple weeks. It usually takes about 2-5 after exposure to the bacteria to start experiencing symptoms. |
| 7 |
Do you cough with strep? |
Other symptoms include sore throat, fever, headache, stomach pain, nausea and vomiting. Cough, hoarseness, red eyes, and runny nose are not seen with Strep throat. These symptoms point more to a viral cause. Scarlet fever rash (fine, red, sandpaper-like rash) is highly suggestive of Strep throat. |
| 8 |
What happens if you don't treat strep A? |
Strep throat accounts for only a small portion of sore throats. If untreated, strep throat can cause complications, such as kidney inflammation or rheumatic fever. Rheumatic fever can lead to painful and inflamed joints, a specific type of rash, or heart valve damage. |
| 9 |
Is strep a hard to get rid of? |
In most cases, antibiotics will quickly wipe out the bacteria causing the infection. In the meantime, try these tips to relieve symptoms of strep throat: Get plenty of rest. Sleep helps your body fight infection. |
| 10 |
What is mistaken for strep throat? |
Sometimes viral illnesses cause a sore throat that may be mistaken for strep throat. These include common cold viruses, coronavirus disease 2019 (COVID-19), influenza (the "flu"), croup, mononucleosis (“mono”), measles, and chickenpox. |
| 11 |
How do I know if I have strep or not? |
Common symptoms of strep throat:
In addition to the standard sore throat and painful swallowing, some other signs and symptoms of strep throat may include: Tender, swollen lymph nodes (glands) in the front of the neck. Red spots on the roof of the mouth or palate. Swollen and red tonsils; white patches on occasion. |

 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!