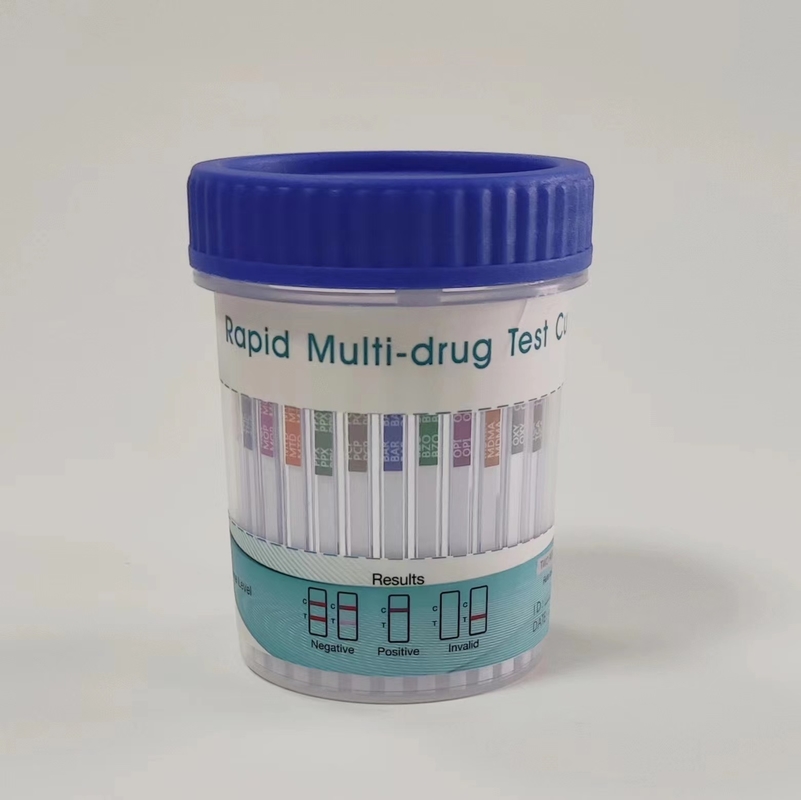
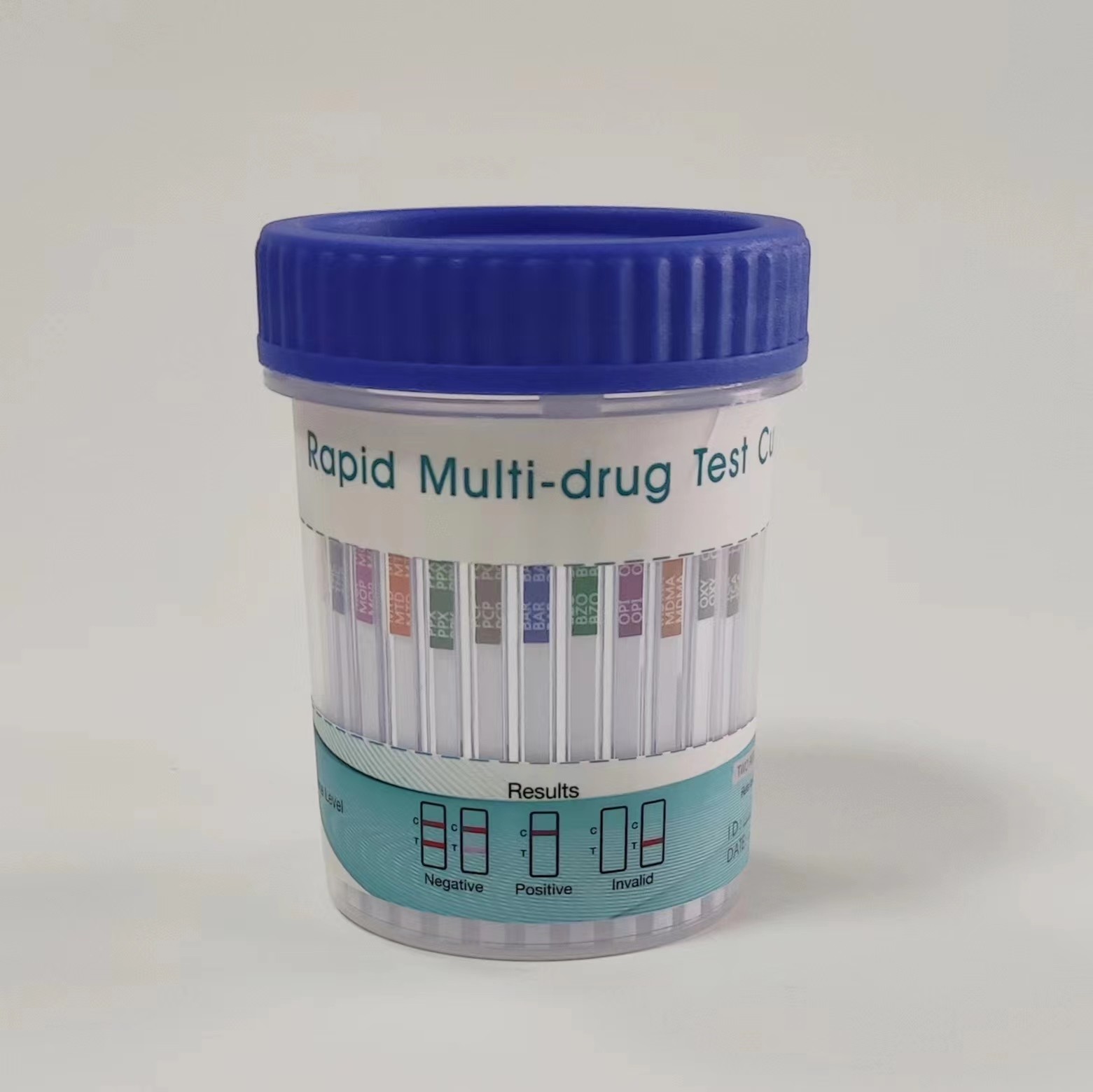
Rapid Multi-Drug 2-12 Test Urine Cup, Rapid Test Cassette
2-12 combination of the following drugs: AMP/ BAR/ BUP/ BZO/ COC/ COT/ ETG/ FYL/ K2/ KET/ MAM/ MDMA
Rapid Multi-Drug 2-12 Test is a rapid, screening test for the qualitative detection of multiple drugs and drug metabolites in human urine at specified cut off levels.
For professional use only. For in vitro diagnostic use only.
【INTENDED USE】
Rapid Multi-Drug 2-12 Test is an immuno-chromatographic assay for the qualitative determination of the presence of drugs listed in the table below.
| Drug(Identifier) |
Calibrator |
Cut-off level |
| Amphetamine (AMP) |
d-Amphetamine |
1000ng/mL |
| Barbiturates (BAR) |
Secobarbital |
300 ng/mL |
| Buprenorphine(BUP) |
Buprenorphine |
10 ng/mL |
| Benzodiazepines (BZO) |
Oxazepam |
300 ng/mL |
| Cocaine (COC) |
Benzoylecgonine |
300 ng/mL |
| Cotinine (COT) |
Cotinine |
200 ng/mL |
| Ethylglucuronide (ETG) |
Ethyl -glucuronide |
500 ng/mL |
| Fentanyl (FYL) |
Fentanyl |
200 ng/mL |
| Synthetic Cannabinoid (K2) |
JWH-018 PantanoicAcid |
50 ng/mL |
| Ketamine (KET) |
Ketamine |
1000ng/mL |
| 6-Aoetylmorphine (MAM) |
6-Aoetylmorphine |
10 ng/mL |
| Methylenedioxymethamphetamine - ecstasy (MDMA) |
3,4-Methylenedioxymethamphetamine HCl (MDMA) |
500 ng/mL |
The test you purchased may test for any combination of drugs listed in the table above. This assay provides only a preliminary analytical test result. Gas Chromatography/Mass spectrometry (GC/MS) is the preferred confirmatory method. Clinical consideration and professional judgment should be applied to any drug of abuse test result, particularly when preliminary positive results are indicated.
【SUMMARY】
Amphetamine (AMP)
Amphetamine and the structurally related “designer” drugs are sympathomimetic amines whose biological effects include potent central nervous system (CNS) stimulation, anorectic, hyperthemic , and cardiovascular properties. They are usually taken orally, intraveneously, or by smoking. Amphetamines are readily absorbed from the gastrointestinal tract and are then either deactivated by the liver or excreted unchanged in the urine . Methamphetamine is partially metabolized to amphetamine and its major active metabolite. Amphetamines increase the heart rate and blood pressure, and suppress the appetite. Some studies indicate that heavy abuse may result in permanent damage to certain essential nerve structural in the brain.The effects of Amphetamines generally last 2-4 hours following use and the drug has a half-life of 4-24 hours in the body. About 30% of amphetamines are excreted in the urine in unchanged form, with the remainder as hydroxylated and deaminated derivatives.It can be detected in the urine for 1 to 2 days after use.
BARBITURATES (BAR)
Barbiturates are central nervous system depressants. They are usually administered orally but are sometimes injected intramuscularly and intravenously. Barbiturates range from short-acting (approximately 15 minutes, such as secobarbital) to long-acting (24 hours or longer, such as Phenobarbital). Short-acting barbiturates are extensively metabolized in the body, while the long-acting ones are secreted primarily unchanged. Barbiturates produce alertness, wakefulness, increased energy, reduced hunger, and an overall feeling of well being. Large doses of Barbiturate could develop tolerance and physiological dependency and lead to its abuse.
Buprenorphine(BUP)
Buprenorphine is a potent analgesic often used in the treatment of opioid addiction. The drug is sold under the trade names Subutex™, Buprenex™, Temgesic™ and Suboxone™, which contain Buprenorphine HCl alone or in combination with Naloxone HCl. Therapeutically, Buprenorphine is used as a substitution treatment for opioid addicts.Substitution treatment is a form of medical care offered to opiate addicts (primarily heroin addicts) based on a similar or identical substance to the drug normally used. In substitution therapy, Buprenorphine is as effective as Methadone but demonstrates a lower level of physical dependence. Concentrations of free Buprenorphine and Norbuprenorphine in urine may be less than 1 ng/ml after therapeutic administration, but can range up to 20 ng/ml in abuse situations.3 The plasma half life of Buprenorphine is 2-4 hours.3 While complete elimination of a single dose of the drug can take as long as 6 days, the window of detection for the parent drug in urine is thought to be approximately 3 days. Substantial abuse of Buprenorphine has also been reported in many countries where various forms of the drug are available. The drug has been diverted from legitimate channels through theft, doctor shopping, and fraudulent prescriptions, and been abused via intravenous, sublingual, intranasal and inhalation routes.
BENZODIAZEPINES (BZO)
Benzodiazepines are a class of drugs that are often therapeutically used as anxiolytics, anti-convulsants and sedative hypnotics. Benzodiazepines manifest their presence by analgesia, drowsiness, confusion, diminished reflexes, lowering of body temperature, respiratory depression, blockade of adrenocortical response, and a decrease in peripheral resistance without an impact on the cardiac index. The major pathways of elimination are the kidneys (urine) and the liver where it is conjugated to glucuronic acid. Large doses of Benzodiazepines could develop tolerances and physiological dependency and lead to its abuse. Only trace amounts (less than 1%) of Benzodiazepines are excreted unaltered in the urine, most of Benzodiazepines in urine is conjugated drug. Oxazepam, a common metabolite of many benzodiazepines, remains detectable in urine for up to one week, which makes Oxazepam a useful marker of Benzodiazepines abuse.
Cocaine (COC)
Cocaine derived from leaves of coca plant, is a potent central nervous system stimulant and a local anesthetic. Among the psychological effects induced by using cocaine are euphoria, confidence and a sense of increased energy, accompanied by increased heart rate, dilation of the pupils, fever, tremors and sweating. Cocaine is excreted in urine primarily as benzoylecgonine in a short period of time.
Cotinine (COT)
Cotinine is the first-stage metabolite of nicotine, a toxic alkaloid that produces stimulation of the autonomic ganglia and central nervous system when in humans. Nicotine is a drug to which virtually every member of a tobacco-smoking society is exposed whether through direct contact or second-hand inhalation. In addition to tobacco, nicotine is also commercially available as the active ingredient in smoking replacement therapies such as nicotine gum, transdermal patches and nasal sprays. In a 24-hour urine, approximately 5% of a nicotine dose is excreted as unchanged drug with 10% as cotinine and 35% as hydroxycotinine; the concentrations of other metabolites are believed to account for less than 5%. While cotinine is thought to be an inactive metabolite, it’s elimination profile is more stable than that of nicotine which is largely urine pH dependent. As a result, cotinine is considered a good biological marker for determining nicotine use.
Ethylglucuronide (ETG)
Ethyl Glucuronide (EtG) is a direct metabolite of alcohol. Presence in urine may be used to detect recent alcohol intake, even after alcohol is no longer measurable. Traditional laboratory methods detect the actual alcohol in the body, which reflects current intake within the past few hours (depending on how much was consumed). The presence of EtG in urine is a definitive indicator that it can be detected in the urine for 3 to 4 days after drinking alcohol, even alcohol is eliminated from the body. Therefore, EtG is a more accurate indicator of the recent intake of alcohol than measuring for the presence of alcohol itself. The EtG test can aid in the diagnosis of drunk driving and alcoholism, which has important significance in the forensic identification and medical examination.
Fentanyl (FYL)
Fentanyl is a potent, synthetic narcotic analgesic with a rapid onset and short duration of action. It is a strong agonist at the μ-opioid receptors. Historically it has been used to treat breakthrough pain and is commonly used in pre-procedures as a pain reliever as well as an anesthetic in combination with a benzodiazepine. It is approximately 80 to 100 times more potent than morphine and roughly 15 to 20 times more potent than heroin.
Synthetic Cannabinoid (K2)
Since 2004, herbal mixtures such as ‘Spice’ are sold in Switzerland, Austria, Germany and other European countries mainly via Internet shops. Although declared as incense, they are smoked as ‘bio-drugs’ by the consumers. In corresponding blogs, drug users reported cannabis-like effects after smoking. These products enjoy great popularity particularly among younger people, as up to now the mixtures are sold in head shops and via internet in many countries without age restriction. JWH-018 was developed and evaluated in basic scientific research to study structure activity relationships related to the cannabinoid receptors. JWH073 has been identified in numerous herbal products, such as “Spice”, “K2”, K3” and others. These products may be smoked for their psychoactive effects.
Ketamine (KET)
Ketamine is a short-acting “dissociative” anesthetic due to its ability to separate perception from sensation. It also has hallucinogenic and painkilling qualities that seem to affect people in very different ways. Ketamine is chemically related to PCP (’Angel Dust’). Ketamine is occasionally administered to people but, more commonly, is used by vets for pet surgery. Generally street K is most often diverted in liquid form from vets’ offices or medical suppliers. Ketamine generally takes 1-5 minutes to take effect. Snorted ketamine takes a little longer at 5-15 minutes. Depending on how much and how recently one has eaten, oral ketamine can take between 5 and 30 minutes to take effect. The primary effects of ketamine last approximately an 30-45 minutes if injected, 45-60 minutes when snorted, and 1-2 hours if used orally. The Drug Enforcement Administration reports that the drug can still affect the body for up to 24 hours.
6-Aoetylmorphine (MAM)
6-Monoacetylmorphine (6-MAM) or 6-acetylmorphine (6-AM) is one of three active metabolites of heroin (diacetylmorphine), the others being morphine and the much less active 3-monoacetylmorphine (3-MAM). 6-MAM is rapidly created from heroin in the body, and then is either metabolized into morphine or excreted in the urine. 6-MAM remains in the urine for no more than 24 hours. So a urine specimen must be collected soon after the last heroin use, but the presence of 6-MAM guarantees that heroin was in fact used as recently as within the last day. 6-MAM is naturally found in the brain, but in such small quantities that detection of this compound in urine virtually guarantees that heroin has recently been consumed.
Methylenedioxymethamphetamine - ecstasy (MDMA)
MDMA belongs to a family of man-made drugs. Its relatives include MDA (methylenedioxyamphetamine), and MDEA (methylenedioxyethylamphetamine). They all share the amphetamine-like effects. MDMA is a stimulant with hallucinogenic tendencies described as an empathogen as it releases mood-altering chemicals, such as cartooning and L-dopa, and may generate feelings of love and friendliness. The adverse effects of MDMA use include elevated blood pressure, hyperthermia, anxiety, paranoia and insomnia. MDMA is administered either by oral ingestion or intravenous injection. The effects of MDMA begin 30 minutes after intake, peak in an hour and last for 2 – 3 hours.
【PRINCIPLE】
Rapid Multi-Drug 2-12 Test is a competitive immunoassay that is used to screen for the presence of various drugs and drug metabolites in urine. It is chromatographic absorbent device in which, drugs within a urine sample, competitively combined to a limited number of drug monoclonal antibody (mouse) conjugate binding sites.
When the test is activated, the urine is absorbed into each test strip by capillary action, mixes with the respective drug monoclonal antibody conjugate, and flows across a pre-coated membrane. When drug within the urine sample is below the detection level of the test, respective drug monoclonal antibody conjugate binds to the respective drug-protein conjugate immobilized in the Test Region (T) of the test strip. This produces a colored Test line in the Test Region (T) of the strip, which, regardless of its intensity, indicates a negative test result.
When sample drug levels are at or above the detection level of the test, the free drug in the sample binds to the respective drug monoclonal antibody conjugate, preventing the respective drug monoclonal antibody conjugate from binding to the respective drug-protein conjugate immobilized in the Test Region (T) of the device. This prevents the development of a distinct colored band in the test region, indicating a preliminary positive result.
To serve as a procedure control, a colored line will appear at the Control Region (C), of each strip, if the test has been performed properly.
【WARNINGS AND PRECAUTIONS】
- Immunoassay for in vitro diagnostic use only.
- Do not use after expiration date.
- The test cassette should remain in the sealed pouch until use.
- The used test cassette should be discarded according to local regulations.
【STORAGE AND STABILITY】
- The kit should be stored at 2-30°C until the expiry date printed on the sealed pouch.
- The test must remain in the sealed pouch until use.
- Keep away from direct sunlight, moisture and heat.
- Do not freeze.
- Care should be taken to protect the components of the kit from contamination. Do not use if there is evidence of microbial contamination or precipitation. Biological contamination of dispensing equipment, containers or reagents can lead to false results.
【SPECIMEN COLLECTION AND PREPRATION】
- Collect urine sample with a clean, dry container. Urine collected at any time of the day may be used.
- For best results, test specimens immediately following collection.
- Urine specimens may be refrigerated (2-8°C) and stored up to forty-eight hours.
- For longer storage, freeze the samples (-20°C or below).
- Bring frozen or refrigerated samples to room temperature before testing.
【OPERATION】
Test must be in room temperature (15ºC to 30ºC)
- Donor collects urine specimen in a urine cup.
- Open the sealed pouch by tearing along the notch. Remove the test cassette from the pouch and place it on a level surface.
- Hold the sample dropper vertically, and add exactly three drops of the urine specimen into the sample well.
- The result should be read at 5 minutes. Do not interpret the result after 10 minutes. See the illustration below.
【TEST LIMITATIONS】
1. This test has been developed for testing urine samples only. No other fluids have been evaluated. DO NOT use this device to test substances other than urine.
2. There is a possibility that technical or procedural errors, as well as interfering substances in the urine specimen may cause erroneous results.
3. Adulterated urine samples may produce erroneous results. Strong oxidizing agents such as bleach (hypochlorite) can oxidize drug analyte. If a sample is suspected of being adulterated, obtain a new sample in a different, unused, cup.
4. This test is a qualitative screening assay. It is not designed to determine the quantitative concentration of drugs or the level of intoxication.
5. A positive result does not indicate level or intoxication, administration route or concentration in urine.
6. A negative result may not necessarily indicate drug-free urine. Negative results can be obtained when drug is present but below the cut-off level of the test.
【QUALITY CONTROL】
A procedural control is included in the test. A line appearing in the Control region (C) is considered an internal procedural control. It confirms sufficient specimen volume, adequate membrane wicking and correct procedural technique.
Control standards are not supplied with this kit. However, it is recommended that positive and negative controls be tested as good laboratory practice to confirm the test procedure and to verify proper test performance. Quality control should be run with each new lot, and every 30 days to check storage stability. Positive and negative control should give the expected results.
Users can commercially obtain control materials (For example from Sigma-Aldrich Corporation).The concentration of drug(s) in positive and negative controls are approximately 50% above and below the cutoff concentration of the assay.
【PERFORMANCE CHARACTERISTICS】
Accuracy
The comparison studies were conducted using Rapid Multi-Drug 2-12 Test and commercially available rapid drugs of abuse tests. The studies were performed on approximately 600 clinical specimens per drug type previous collected from the clinical settings. Results were as follows:
| Format |
% Agreement with Predicate Test |
Format |
% Agreement with Predicate Test |
| AMP |
Positive |
100.00% |
K2 |
Positive |
100.00% |
| Negative |
99.31% |
Negative |
99.65% |
| BAR |
Positive |
100% |
KET |
Positive |
98.65% |
| Negative |
99.30% |
Negative |
99.05% |
| BUP |
Positive |
100.00% |
MAM |
Positive |
97.62% |
| Negative |
98.78% |
Negative |
99.28% |
| BZO |
Positive |
100% |
MDMA |
Positive |
100% |
| Negative |
99.20% |
Negative |
99.50% |
| COC |
Positive |
100.00% |
ETG
|
Positive |
97.83% |
| Negative |
99.66% |
Negative |
99.46% |
| COT |
Positive |
98.55% |
FYL
|
Positive |
100.00% |
| Negative |
99.44% |
Negative |
99.64% |
Specificity and cross reactivity
To test the specificity and cross reactivity of the test, the test device was used to test various drugs, drug metabolites and other components of the same class that are likely to be present in urine. All the components were added to drug-free normal human urine. The following structurally related compounds produced positive results with the test when tested at levels equal to or greater than the concentrations listed below.
| Compound |
Response equivalent
to cutoff in ng/mL
|
Compound |
Response equivalent
to cutoff in ng/mL
|
| Amphetamine (AMP) |
|
6-Aoetylmorphine (MAM) |
|
| d-Amphetamin |
1,000 |
6-Monoacetylmorphine(6-MAN) |
10 |
| d.l-Amphetamine |
2,500 |
Morphine |
100 |
| 1-Amphetamine |
50,000 |
Codeine |
100 |
| (+/-) 3,4-methylenedioxyamphetamine (MDA) |
2,000 |
Hydrocodone |
1000 |
| Barbiturates (BAR) |
|
Hydromorphine |
1000 |
| Secobarbital |
300 |
Morphine 3-b-D-glucuronide |
50 |
| Amobarbital |
500 |
Methylenedioxymethamphetamine - ecstasy (MDMA) |
|
| Alphenol |
150 |
D-Amphetamine |
>100000 |
| Aprobarbital |
200 |
(+/-)3,4-Methylenedioxymethamphetamine (MDMA) |
500 |
| Butabarbital |
75 |
3,4-methylenedioxyamphetamine (MDA) |
2200 |
| Butalbital |
1,500 |
3,4-Methylenedioxyethylamphetamine (MDEA) |
240 |
| Butethal |
100 |
Ketamine (KET) |
|
| Cyclopentobarbital |
600 |
Ketamine |
1,000 |
| Pentobarbital |
700 |
Norketamine |
3,000 |
| Phenobarbital |
300 |
Methoxy-amphetamine |
12,500 |
| Buprenorphine(BUP) |
|
Promethazine |
25,000 |
| Buprenorphine |
10 |
4 - hydroxyphenyl cyclohexyl piperidine |
50,000 |
| Norbuprenorphine |
20 |
Synthetic Cannabinoid (K2) |
|
| Buprenorphine 3-D-glucuronide |
15 |
JWH-018 Pentanoic Acid metabolite |
50 |
| Norbuprenorphine 3-D-glucuronide |
200 |
JWH-073 Butanoic Acid metabolite |
50 |
| Benzodiazepines (BZO) |
|
JWH-018 N-4-hydroxypentyl |
16,000 |
| Oxazepam |
300 |
JAM2201 N-Pentanoic Acid metabolite |
400 |
| Alprazolam |
200 |
JWH398 N-Pentanoic Acid metabolite |
800 |
| α-Hydroxyalprazolam |
1100 |
JWH-210 N-(5-carboxypentyl) metabolite |
5,000 |
| Bromazepam |
1000 |
JWH-073 3-hydroxypentyl metabolite |
5,000 |
| Chlordiazepoxide |
2000 |
JWH-019 5-hydroxypentyl metabolite |
80,000 |
| Clobazam |
100 |
JWH-018 5-hydroxypentyl metabolite |
90,000 |
| Clonazepam |
800 |
JWH-073 4-hydroxypentyl metabolite |
80,000 |
| Clorazepate |
200 |
Cocaine (COC) |
|
| Delorazepam |
1600 |
Benzoylecgonine |
300 |
| Diazepam |
200 |
Cocaine |
800 |
| Estazolam |
1000 |
Cocaethylene |
12,500 |
| Flunitrazepam |
350 |
Ecgonine HCl |
35,000 |
| Lorazepam |
1200 |
Cotinine (COT) |
|
| Midazolam |
2500 |
(-)-Cotinine |
200 |
| Nitrazepam |
100 |
(-)-Nicotine |
6,250 |
| Nordiazepam |
400 |
Ethylglucuronide (ETG) |
|
| Temazepam |
120 |
Ethyl-β-D-glucuronide |
500 |
| Triazolam |
1000 |
Ethyl-β-D-glucuronide-D5 |
500 |
| Fentanyl (FYL) |
|
|
|
| Fentanyl |
200 |
|
|
Interfering substances
Clinical urine samples may contain substances that could potentially interfere with the test. The following compounds were added to drug-free urine urine with the concentration 50% below and above the cutoff, respectively. All potential interfering substances were added at a concentration of 100µg/mL. The urine specimens were tested with the test device. None of the urine samples showed any deviation from the expected results.
| Acetaminophen |
Chlorothiazide |
Estrone-3-sulfate |
Isoxsuprine |
Oxymetazoline |
Sulindac |
| Acetophenetidin |
Chlorpheniramine |
Ethyl-p-aminobenzoate |
Ketoprofen |
Oxytetracycline |
Tetracycline |
| Amoxicillin |
d,l-Chlorpromazine |
Erythromycin |
Labetalol |
Papaverine |
Tetrahydrozoline |
| Ampicillin |
Cholesterol |
Fenoprofen |
Lisinopril |
Penicillin-G |
Thiamine |
| Aspirin |
Clonidine |
Flucloxacillin |
Loperamide |
Pentazocine |
Thioridazine |
| Atenolol |
Cimetidine |
Fluoxetine |
Meperidine |
Perphenazine |
d, l-Thyroxine |
| Atorvastatin |
Citalopram |
Furosemide |
Meprobamate |
Phenelzine |
Tolbutamine |
| Azlocillin |
Cortisone |
Gentisic acid |
Methoxyphenamine |
Prednisolone |
Tolbutamide |
| Benzilic acid |
Creatinine |
Hemoglobin |
Methylphenidate |
Prednisone |
Trifluoperazine |
| Benzylpenicillin |
Deoxycorticosterone |
Hydralazine |
Nadolol |
d,l-Propanolol |
Tryptamine |
| Benzoic acid |
Dexamethasone |
Hydrochlorothiazide |
Nalidixic acid |
d-Pseudoephedrine |
Uric acid |
| Bilirubin |
Dextromethorphan |
Hydrocortisone |
Naproxen |
Quinacrine |
Verapamil |
| Benzydamine |
Diclofenac |
o-Hydroxyhippuric acid |
Niacinamide |
Quinine |
Zomepirac |
| Caffeine |
Diflunisal |
p-Hydroxytyramine |
Nifedipine |
Quindine |
|
| Carbamazepine |
Digoxin |
Ibuprofen |
Norethindrone |
Ranitidine |
|
| Cephalexin |
Diphenhydramine |
Indomethacin |
d,l-Octopamine |
Salicylic acid |
|
| Chloralhydrate |
Ephedrine |
Iproniazid |
Oxalic acid |
Serotonin |
|
| Chloramphenicol |
β-Estradiol |
d,l-Isoproterenol |
Oxolinic acid |
Sulfamethazine |
|


 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!