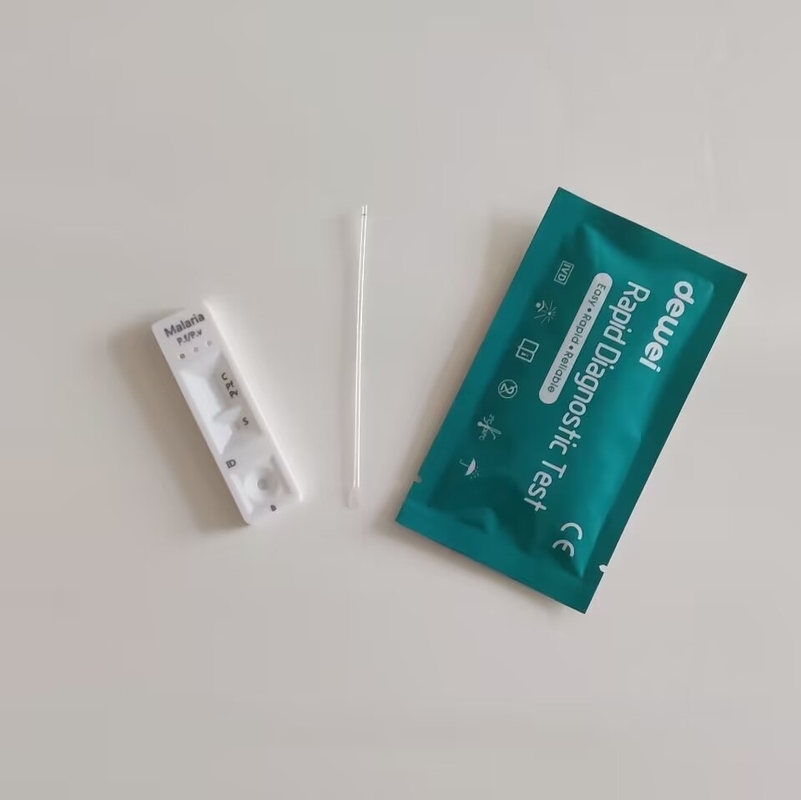
One Step Malaria Plasmodium Falciparum P.f. Antigen Rapid Test Diagnostic Kit
INTENDED USE
The Malaria plasmodium falciparum (P.f.) Rapid Test (Whole Blood) is a rapid chromatographic immunoassay for the qualitative detection of circulating antigens of Plasmodium falciparum in whole blood.
INTRODUCTION
Malaria is caused by a protozoan which invades human red blood cells.1 Malaria is one of the world's most prevalent diseases. According to the WHO, the worldwide prevalence of the disease is estimated to be 300-500 million cases and over 1 million deaths each year. Most of these victims are infants, young children. Over half of the world's population lives in malarious areas. Microscopic analysis of appropriately stained thick and thin blood smears has been the standard diagnostic technique for identifying malaria infections for more than a century.2 The technique is capable of accurate and reliable diagnosis when performed by skilled microscopists using defined protocols. The skill of the microscopist and use of proven and defined procedures, frequently present the greatest obstacles to fully achieving the potential accuracy of microscopic diagnosis. Although there is a logistical burden associated with performing a time-intensive, labor-intensive, and equipment-intensive procedure such as diagnostic microscopy, it is the training required to establish and sustain competent performance of microscopy that poses the greatest difficulty in employing this diagnostic technology.
The Malaria P.f. Rapid Test (Whole Blood) is a rapid test to qualitatively detect the presence of the P.f. antigen.
PRINCIPLE
The Malaria P.f. Rapid Test Device (Whole Blood) is a qualitative, membrane based immunoassay for the detection of P.f. antigen in whole blood. The membrane is pre-coated with P.f. antibody. During testing, the whole blood specimen reacts with the dye conjugate, which has been pre-coated in the test strip. The mixture then migrates upward on the membrane chromatographically by capillary action and reacts with P.f. antibody on the membrane on the test line. If the specimen contains P.f. antigen, a colored line will appear in the test region. The absence of the colored line in test region indicates that the specimen does not contain P.f. antigen. To serve as a procedure control, a colored line will always appear in the control region indicating that proper volume of specimen has been added and membrane wicking has occurred.
MAIN CONTENTS
Rapid Test Cassette in Pouch
Buffer
Disposable dropper
Package insert
SRORAGE
Store as packaged in the sealed pouch at room temperature or refrigerated (2-30°C).
The test is stable through the expiration date printed on the sealed pouch.
The test must remain in the sealed pouch until use.
DO NOT FREEZE.
Do not use beyond the expiration date.
OPERATION PROCEDURE
Allow the test device, specimen, buffer, and/or controls to equilibrate to room temperature (15-30°C) prior to testing.
1. Remove the test device from the foil pouch and use it as soon as possible. Best results will be obtained if the assay is performed within one hour.
2. Place the test device on a clean and level surface. Transfer the specimen by a pipette or a dropper:
3. To use a Pipette: Transfer 10 mL of whole blood to the specimen well (S) of the test device, then add 3 full drops of buffer (approximately 120 mL) and start the timer. Avoid trapping air bubbles in the specimen well (S). See illustration ① below.
4. To use a Disposable Specimen Dropper: Hold the dropper vertically, draw the specimen up to the Fill Line as shown in illustration ② below (approximately 10mL). Transfer the specimen to the specimen well (S) of the test device, then add 3 full drops of buffer (approximately 120 uL) and start the timer. Avoid trapping air bubbles in the specimen well (S)
5. Wait for the red line(s) to appear. The result should be read at 15 minutes. Do not interpret the result after 20 minutes.
INTERPRETATION
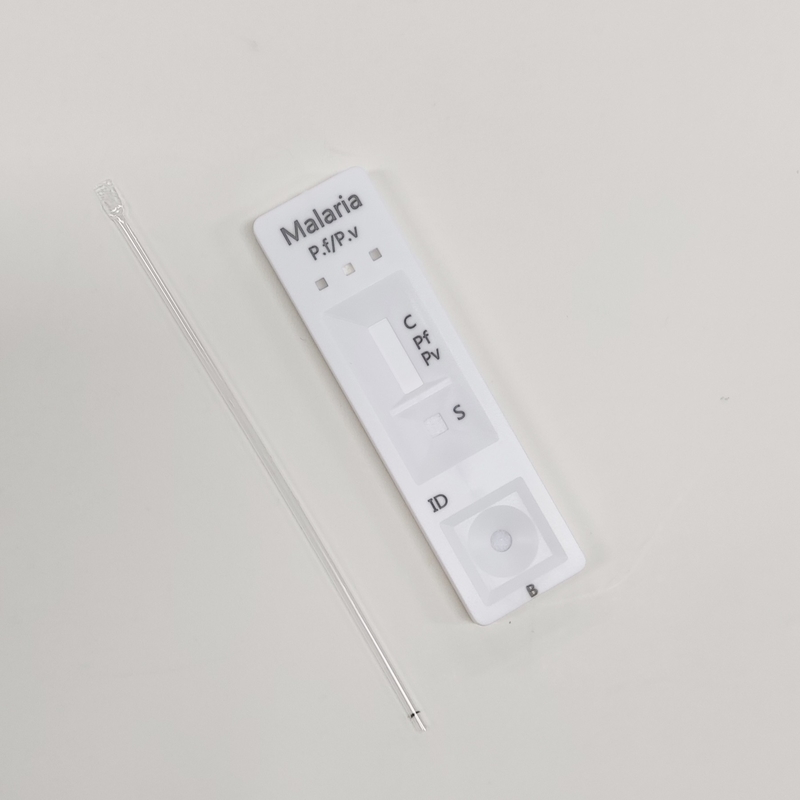
POSITIVE: Two colored bands appear on the membrane. One band appears in the control region (C) and another band appears in the test region (T).
NEGATIVE: Only one colored band appears, in the control region (C). No apparent colored band appears in the test region (T).
INVALID: Control band fails to appear. Results from any test which has not produced a control band at the specified read time must be discarded. Please review the procedure and repeat with a new test. If the problem persists, discontinue using the kit immediately and contact your local distributor.
Note:
- The intensity of color in the test region (T) may vary depending on the concentration of analytes present in the specimen. Therefore, any shade of color in the test region should be considered positive. Note that this is a qualitative test only, and cannot determine the concentration of analytes in the specimen.
- Insufficient specimen volume, incorrect operating procedure or expired tests are the most likely reasons for control band failure.
For details, please contact Dewei staff for Instruction Manual!


 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!