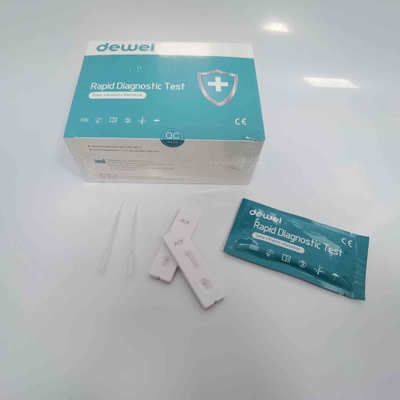
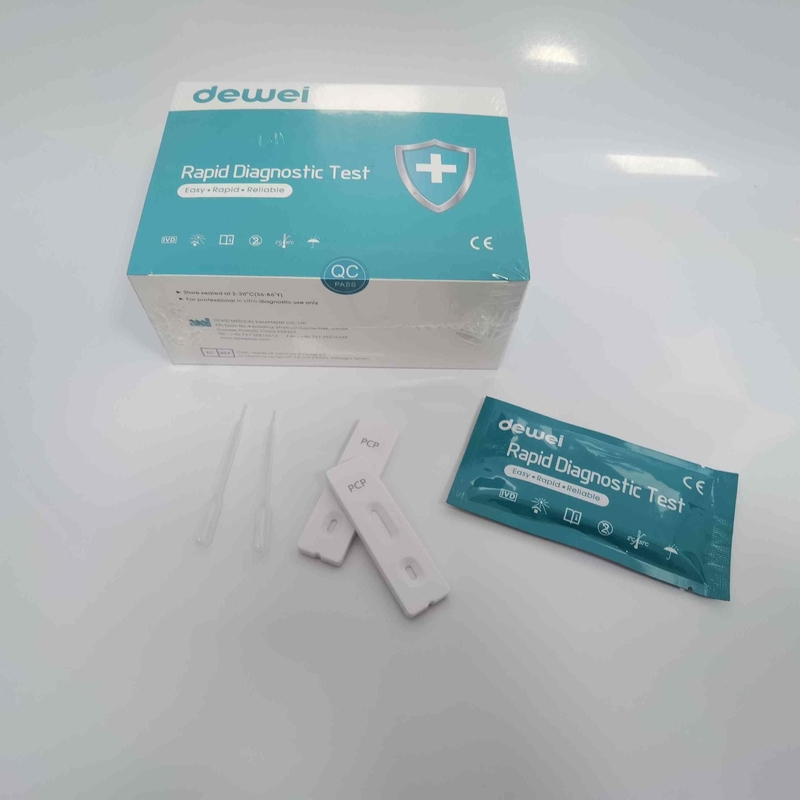
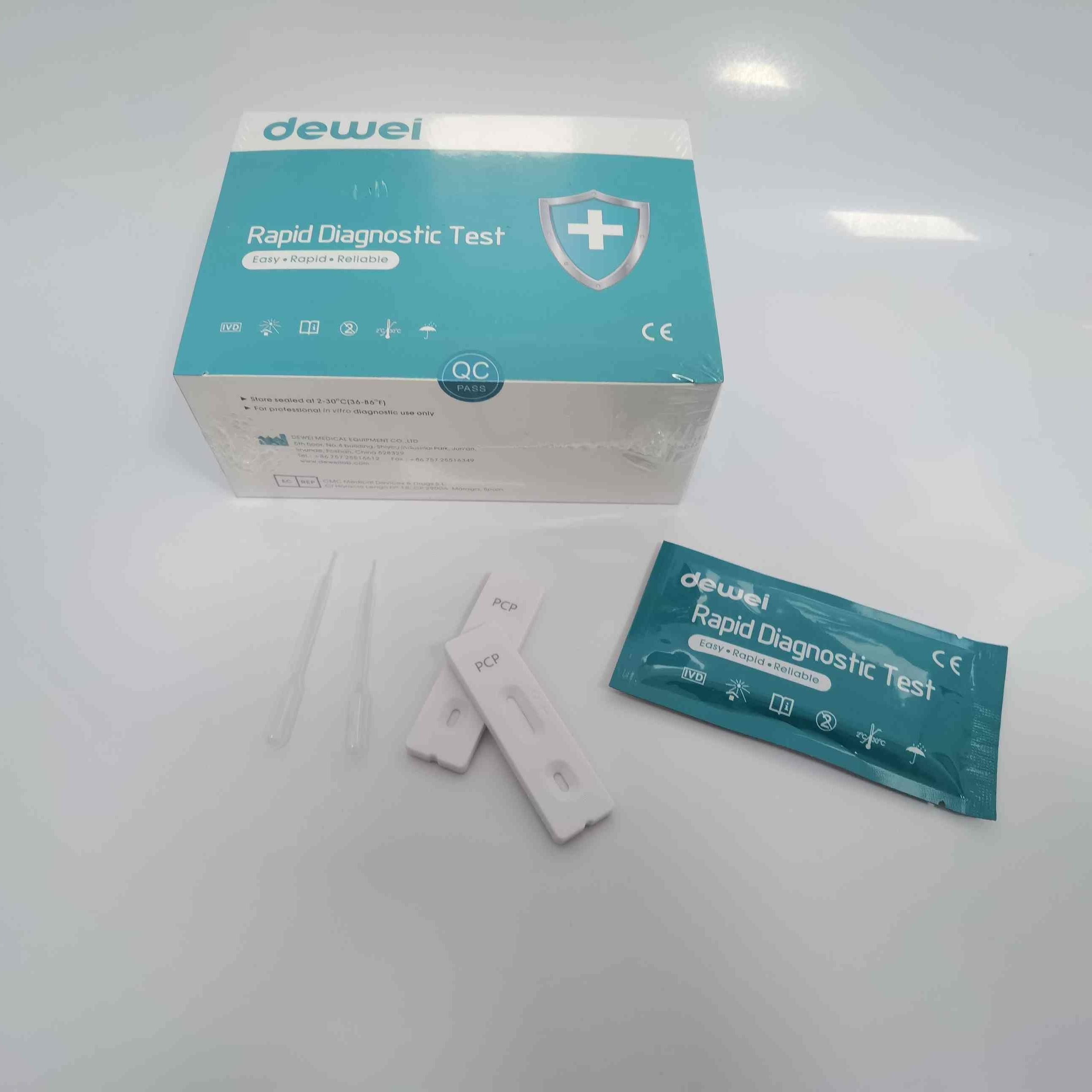
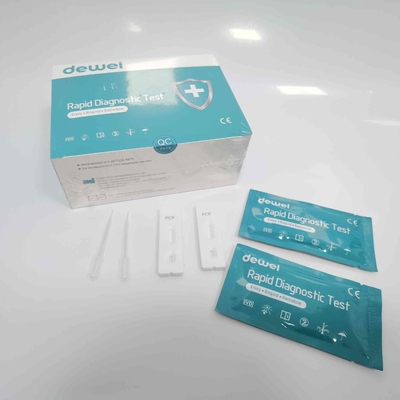
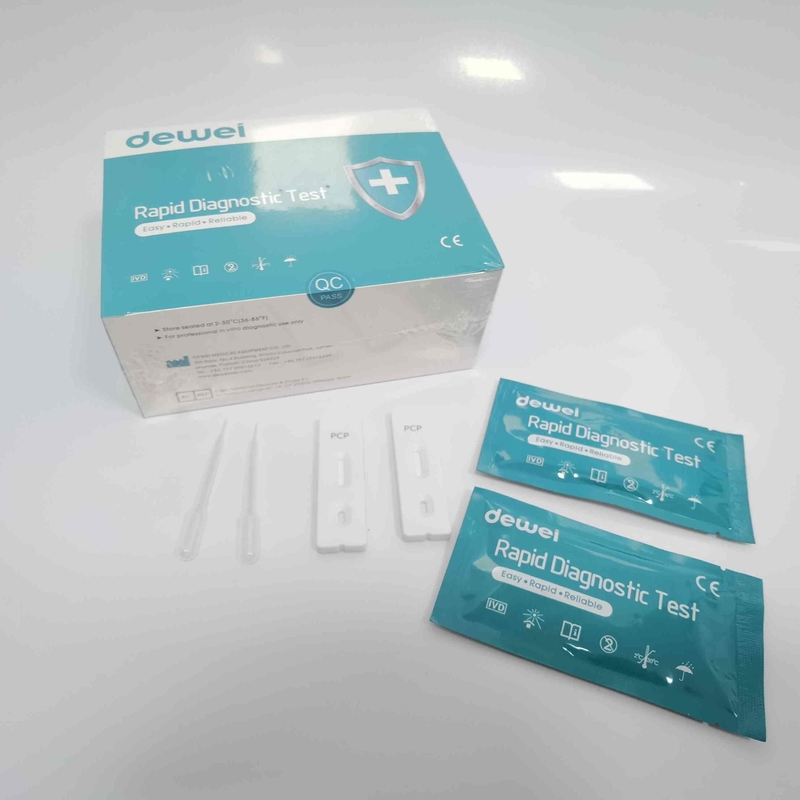
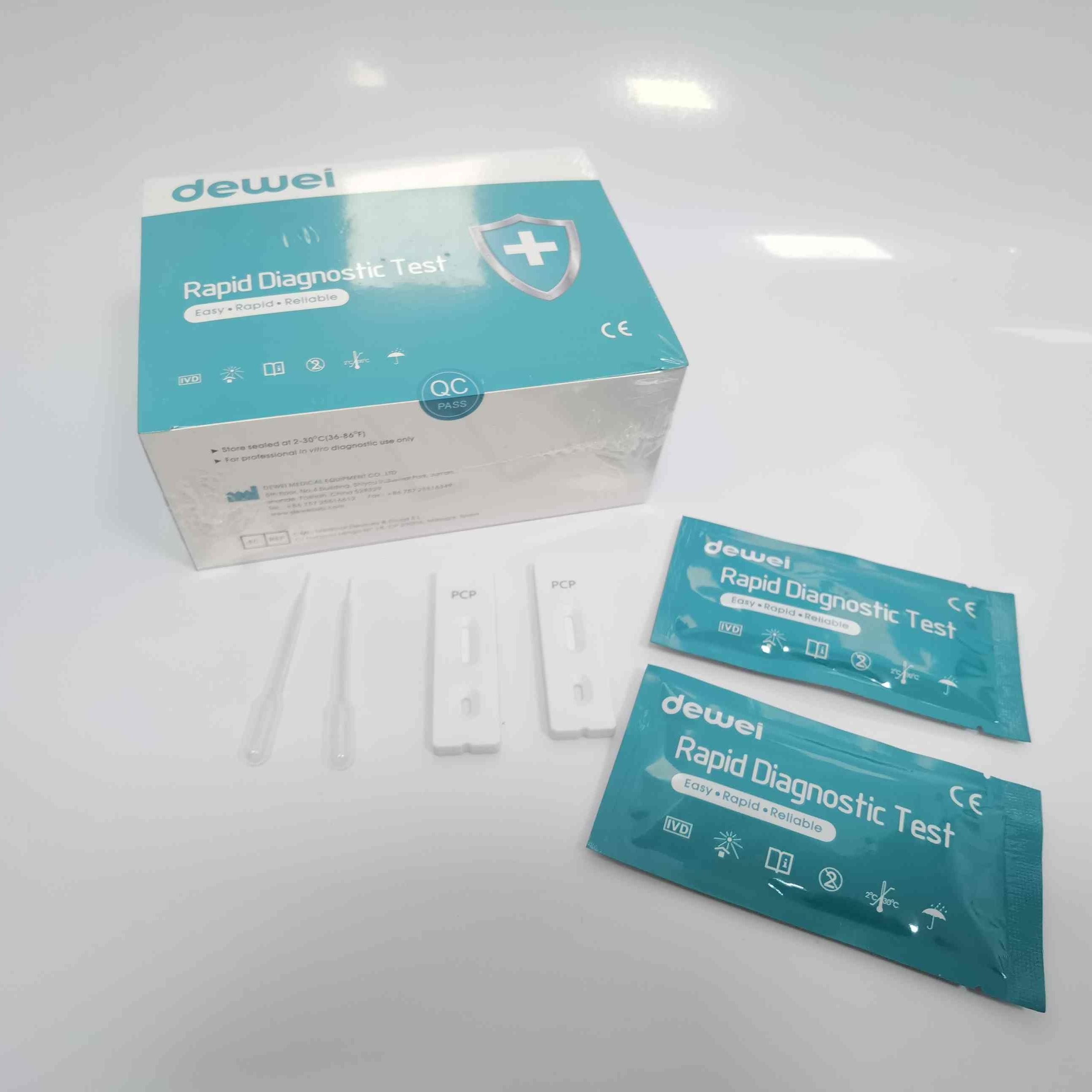
CE PCP Rapid Test Kit Drug of Abuse Rapid Test Cassette for Urine Sample
PCP Rapid Test is a rapid, screening test for the qualitative detection of PCP an metabolites in human urine at specified cut oflevels.
For professional use only.
For in vitro diagnostic use only.
【INTENDED USE】
PCP Rapid Test is an immuno-chromatographic assay for the qualitative determination of the presence of drugs listed in the table below.
| Drug(Identifier) |
Calibrator |
Cut-off level |
| PCP |
PCP |
25 ng/mL |
This assay provides only a preliminary analytical test result. Gas Chromatography/Mass spectrometry (GC/MS) is the preferred confirmatory method. Clinical consideration and professional judgment should be applied to PCP of abuse test result, particularly when preliminary positive results are indicated.
【SUMMARY】
PCP, commonly known as PCP or “angel dust” is used primarily as recreational drug due to its hallucinogenic effects. It is generally self-administered by intravenous injection or by inhalation and concentrates fastest in fatty tissues and the brain. The effects of PCP are very much dose related. Small amounts of PCP are central nervous system stimulants that produce alertness, wakefulness, increased energy, increased heat rate, and decreased sense of pain and touch, and an overall feeling of well being. Large doses of PCP can result in death due to convulsions, heart and lung failure and coma . Large repeated doses of PCP could develop tolerances and physiological dependency and lead to its abuse. PCP can be found in urine within 4 to 6 hours after use and will remain in urine for 7 to 14 days. PCP is excreted in the urine as an unchanged drug (4% to 19%) and conjugated metabolites (25% to 30%).
【PRINCIPLE】
PCP Rapid Test is a competitive immunoassay that is used to screen for the presence of PCP and metabolites in urine. It is chromatographic absorbent device in which, drugs within a urine sample, competitively combined to a limited number of drug monoclonal antibody (mouse) conjugate binding sites.
When the test is activated, the urine is absorbed into each test strip by capillary action, mixes with the respective drug monoclonal antibody conjugate, and flows across a pre-coated membrane. When drug within the urine sample is below the detection level of the test, respective drug monoclonal antibody conjugate binds to the respective drug-protein conjugate immobilized in the Test Region (T) of the test strip. This produces a colored Test line in the Test Region (T) of the strip, which, regardless of its intensity, indicates a negative test result.
When sample drug levels are at or above the detection level of the test, the free drug in the sample binds to the respective drug monoclonal antibody conjugate, preventing the respective drug monoclonal antibody conjugate from binding to the respective drug-protein conjugate immobilized in the Test Region (T) of the device. This prevents the development of a distinct colored band in the test region, indicating a preliminary positive result.
To serve as a procedure control, a colored line will appear at the Control Region (C), of each strip, if the test has been performed properly.
【WARNINGS AND PRECAUTIONS】
- Immunoassay for in vitro diagnostic use only.
- Do not use after expiration date.
- The test should remain in the sealed pouch until use.
- The used test should be discarded according to local regulations.
【CONTENTS】
- Drug Test.
- Desiccant
- Leaflet with instruction for use.
【STORAGE AND STABILITY】
- The kit should be stored at 2-30°C until the expiry date printed on the sealed pouch.
- The test must remain in the sealed pouch until use.
- Keep away from direct sunlight, moisture and heat.
- Do not freeze .
- Care should be taken to protect the components of the kit from contamination. Do not use if there is evidence of microbial contamination or precipitation. Biological contamination of dispensing equipment, containers or reagents can lead to false results.
【OPERATION】
Test must be in room temperature ( 15ºC to 30ºC)
- Donor collects urine specimen in a urine cup.
- Open the sealed pouch by tearing along the notch. Remove the test from the pouch and place it on a level surface.
- Hold the sample dropper vertically, and add exactly three drops of the urine specimen into the sample well.
- The result should be read at 5 minutes. Do not interpret the result after 10 minutes. See the illustration below.
【INTERPRETATION OF RESULTS】
Preliminary positive (+)
Only one colored band appears, in the control region (C). No colored band appears in the test region (T).
Negative (-)
Two colored bands appear on the membrane. One band appears in the control region (C) and another band appears in the test region (T).
Invalid
Control band fails to appear. Results from any test which has not produced a control band at the specified read time must be discarded. Please review the procedure and repeat with a new test. If the problem persists, discontinue using the
kit immediately and contact your local distributor.

 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!