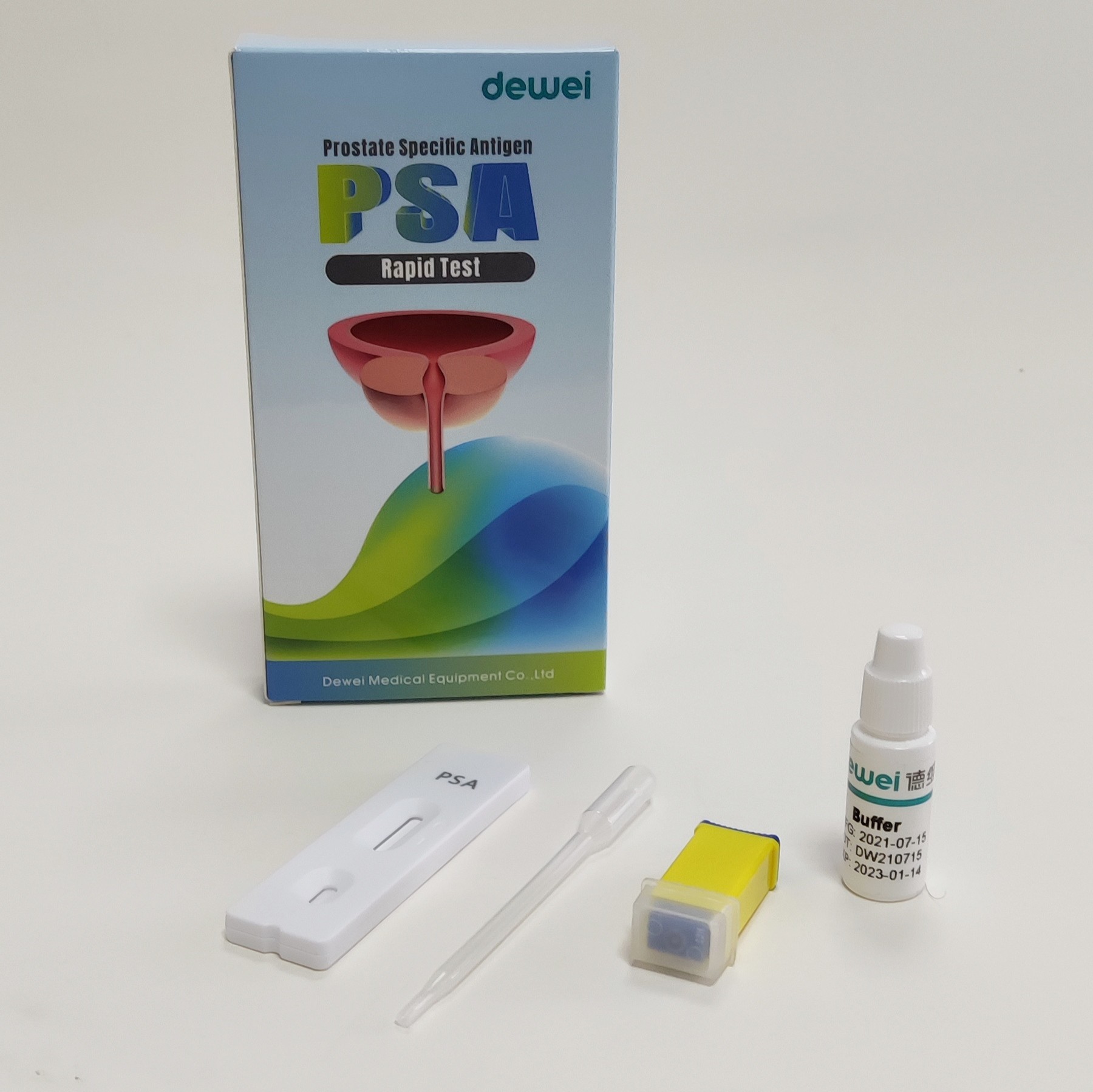
CE ISO Rapid Test Kit Prostate Specific Antigen (PSA) Rapid Test Tumor Marker
[INTENDED USE]
The Prostate Specific Antigen (PSA) is a rapid visual immunoassay for the qualitative presumptive detection of prostate specific antigens in human whole blood, serum, or plasma specimens. This kit is intended for use as an aid in the diagnosis of prostate cancer.
[CONTENTS]
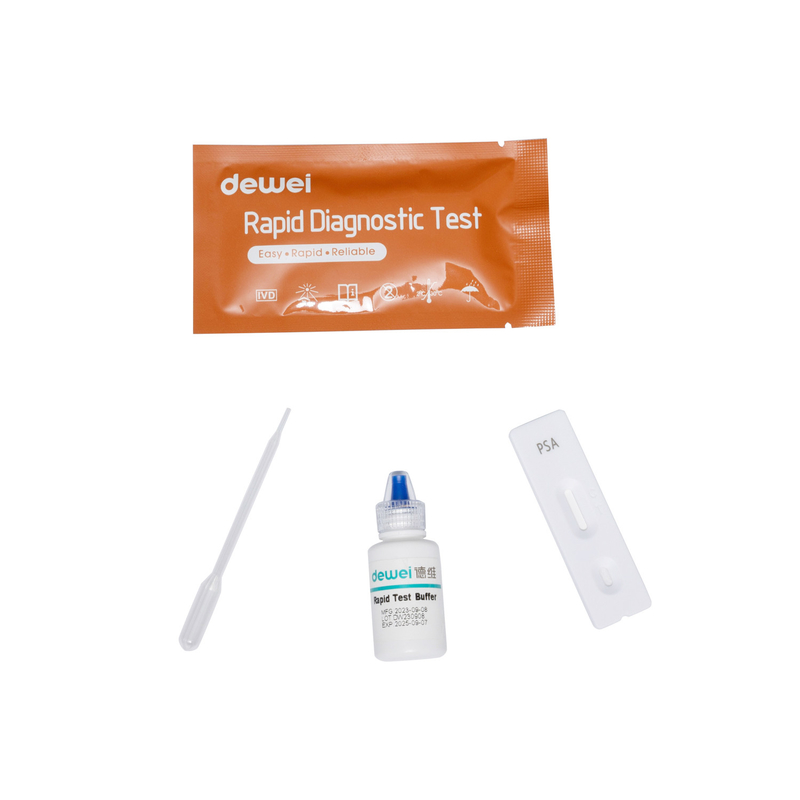
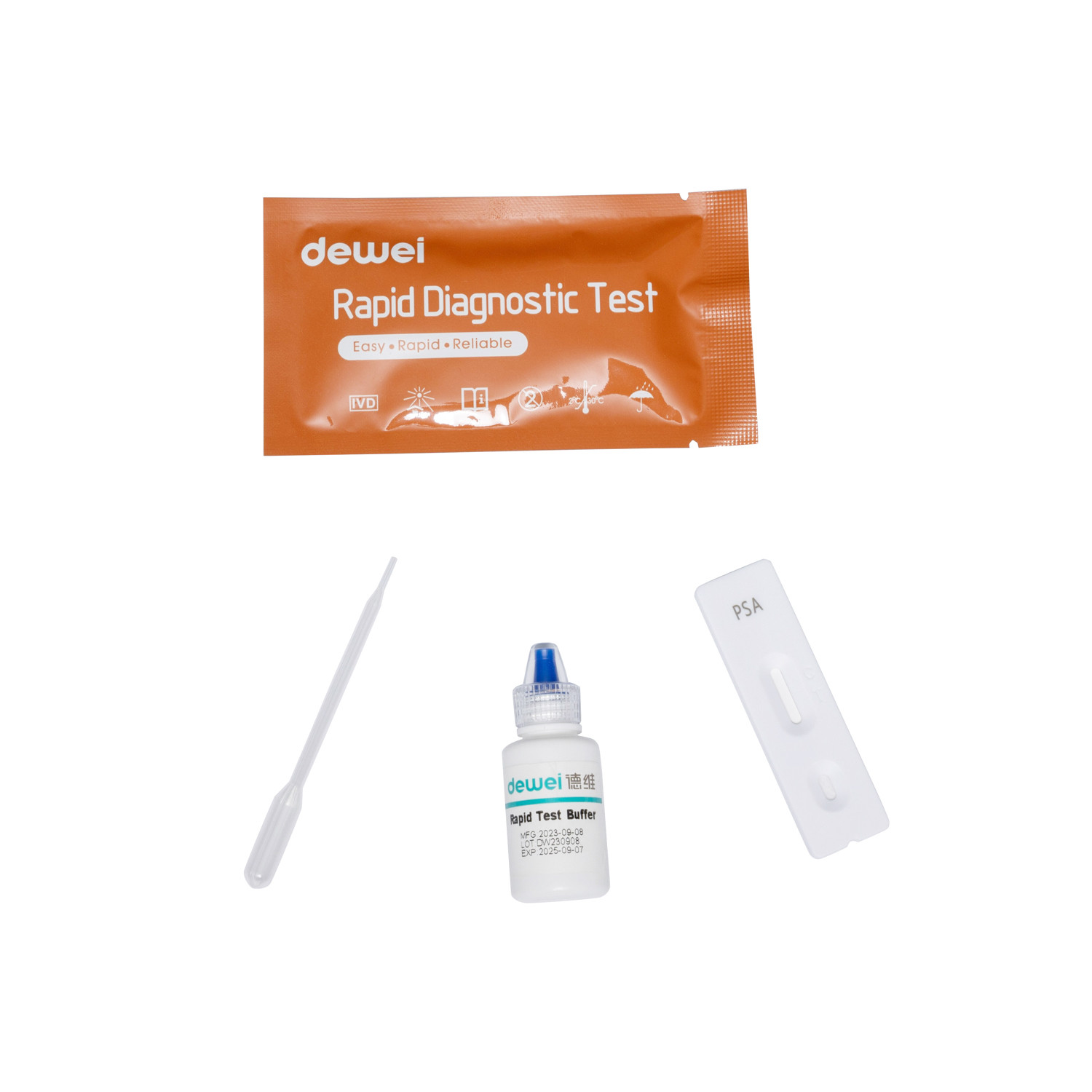
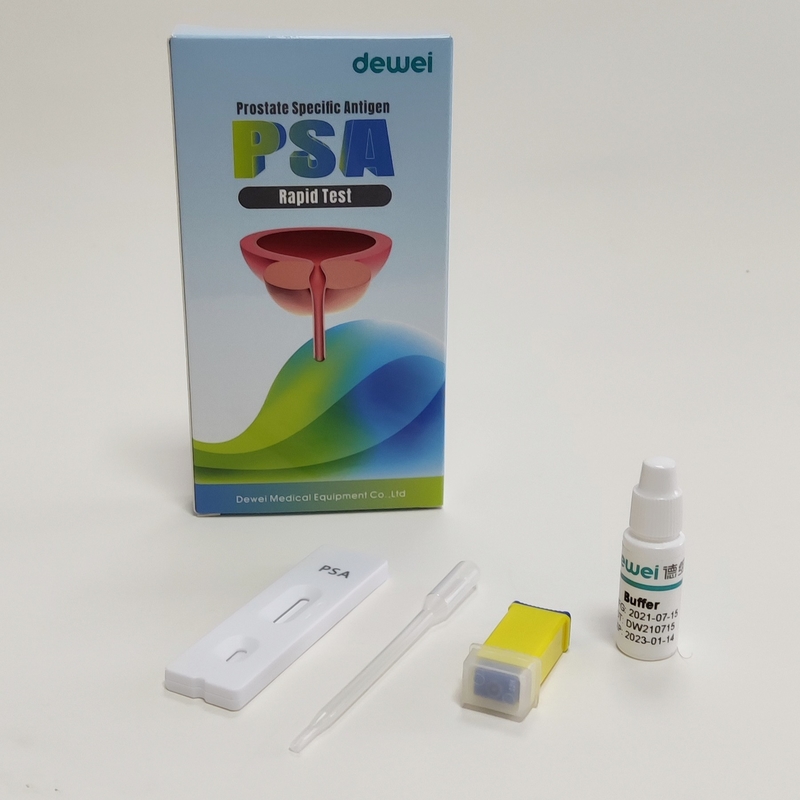
Rapid test cassette with desiccant.
• Buffer
• Pipette
• Package insert
[OPERATION]
Bring tests, specimens, buffer and/or controls to room temperature (15- 30°C) before use.
1. Remove the test from its sealed pouch, and place it on a clean, level surface. Label the device with patient or control identification. For best results, the assay should be performed within one hour.
2. Transfer 1 drops of serum/plasma to the specimen well (S) of the device with the provided disposable pipette, then add 1 drop of buffer, and start the timer.
OR
Transfer 2 drops of whole blood to the specimen well (S) of the device with the provided disposable pipette, then add 1 drop of buffer, and start the timer.
OR
Allow 2 hanging drops of fingerstick whole blood to fall into the center of the specimen well (S) of the test device, then add 1 drop of buffer, and start the timer.
Avoid trapping air bubbles in the specimen well (S), and do not add any solution to the result area.
As the test begins to work, color will migrate across the membrane. 3. Wait for the colored band(s) to appear. The result should be read at 10 minutes. Do not interpret the result after 20 minutes.
[STORAGE AND STABILITY]
1,The kit should be stored at 2-30°C until the expiry date printed on the sealed pouch.
2,The test must remain in the sealed pouch until use. Keep away from direct sunlight, moisture and heat.
3,Do not freeze.
4,Care should be taken to protect the components of the kit from contamination.
5,Do not use if there is evidence of microbial contamination or precipitation. Biological contamination of dispensing equipment, containers or reagents can lead to false results.
[ABOUT US]
DEWEI Medical Equipment Co., Ltd is a leading and professional manufacturer of IVD products,it is a high-tech enterprise specialized in developing, manufacturing and marketing of Clinical Laboratory analyzer & reagents, such as Hematology Analyzer & Reagents, Virus RNA Preservation & Extraction Kits, Urine Sediment Reagents and Washing Solutions.
DEWEI products are widely used in hospital clinics, disease control centers, blood banks, veterinary clinics, scientific research center and college laboratories.
Qulity is primary concern, DEWEI team is strict on quality control systems thus our products have CE, ISO, CFDA and other international standards certificates.
The view of DEWEI is to be the most professional and respected enterprise in IVD field, DEWEI will go on providing best service to you by the outstanding R&D and cost-effective products.

 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!