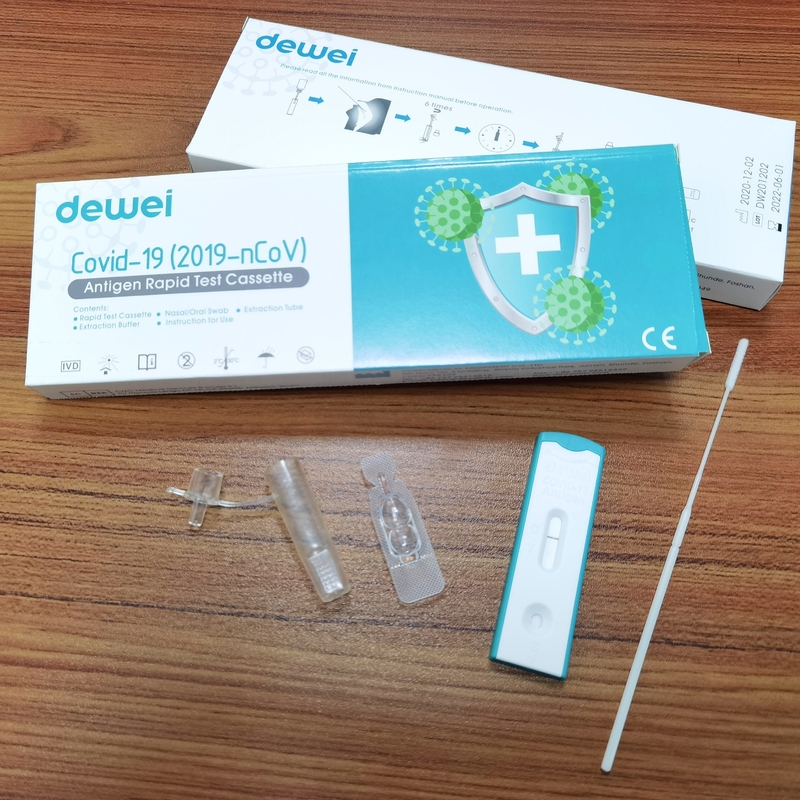
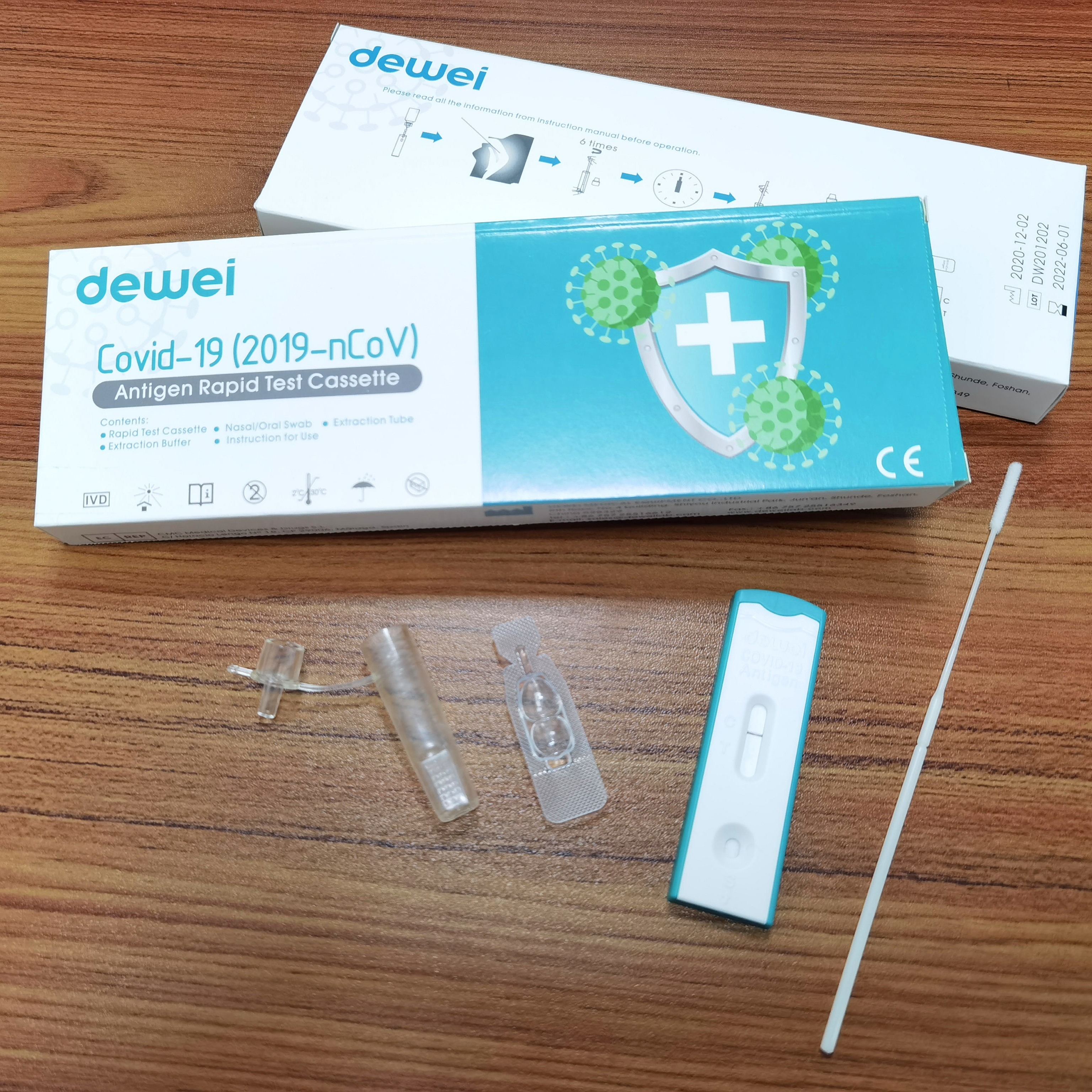
Antigen Sample Swab POCT Rapid Test Cassette Strip 1test/Box 25tests/Box for Covid-19
Individual Pack for Home Used Self Test!
² Easy: One Step
² Fast: 15 Minutes
² Cheap: Lower cost with high efficiency
² Convenient: Throat/Nasal swab sample
【MAIN CONTENTS OF ANTIGEN TEST】
• Rapid Test Cassette with desiccant;
• Extraction Tube;
• Extraction Reagent;
• Instructions for use;
• Nasal/Oral Swab;
• Working Holder.
【STORAGE & STABILITY OF ANTIGEN TEST】
• Store at 39 ~ 86 º F (4 ~ 30 º C) in the sealed pouch up to the expiration date.
• Keep away from direct sunlight, moisture and heat.
• DO NOT FREEZE.
【SAMPLE COLLECTION OF ANTIGEN TEST】
This product can be equipped with disposable sampling swabs, sampling method is as follows:
- Nasal Sampling: Gently insert the swab head into the nasal cavity and scrape the cavity wall. Immerse the swab head into sample solution and
All specimens should be tested as soon as early they are prepared. If necessary, they may be stored at 2-8°C for up to 24 hours or at -20°C for longer periods. Restore the sample to room temperature before test.
What is the difference between an antigen test and a molecular test?
An antigen test detects specific proteins on the surface of the virus. These tests are quicker and less expensive, but have a higher chance of missing an active infection, according to the FDA.
A molecular (PCR or polymerase chain reaction) test detects the virus’s genetic material. These tests require a more complex technology to get results, and it usually takes a day or two to get those results (depending on lab capacity, results may take up to a week).
If an antigen test shows a negative result and you have reason to believe you may have the virus (because of symptoms or exposure), your doctor may order a molecular test to confirm the results.
Which tests are more accurate?
No test is 100% accurate, but the molecular tests are considered to be more accurate than the antigen tests, according to available research.
According to the FDA, an antigen test cannot “definitively rule out active coronavirus infection,” but positive results are “highly accurate” (negative results may require a confirmation test).
Harvard Medical School wrote in August that the reported rate of false negatives with molecular testing is as low as 2% and as high as 37%. A molecular test using a deep nasal swab will have fewer false negative results than samples from throat swabs or saliva, they say.
For antigen testing, Harvard noted that the reported rate of false negative results can be as high as 50%, but that the FDA has granted emergency use authorization for a more accurate antigen test.
In general, some of the issues that may affect the accuracy of a test include ineffective swabbing, contamination or mishandling of the sample, or problems with the testing chemicals, according to the FDA.
If antigen tests are less accurate, why would we use them?
Experts say the value in the rapid antigen tests is in the frequency of the testing rather than the accuracy, with repeated testing recommended. According to Science, getting a false negative in an antigen test two or three times in a row is rare. This strategy lines up with the NPR report mentioned above, which would employ daily testing of those in high-risk areas.
If an individual has been vaccinated or immunized for COVID-19, do they still need to be tested prior to visiting a long-term care home?
Yes, the testing requirements of the Minister’s Directive continue to apply to individuals who have been vaccinated, in addition to continuing to follow public health measures including masking, physical distancing, hand hygiene, and symptom screening. This includes active screening on entry to the long-term care home for symptoms and exposures for COVID-19, including temperature checks, attesting to not be experiencing any of the typical and atypical symptoms of COVID-19 (in accordance with Directive #3 issued by the Chief Medical Officer of Health).
Do individuals who test positive on the rapid antigen test need to be confirmed with lab-based PCR testing?
A positive test result on the rapid antigen test should be considered a preliminary positive and requires a confirmatory laboratory-based PCR test. The following actions should be taken:
1. Counsel individual that the result is preliminary positive and PCR confirmation is required.
2. Issue guidance to return home and self-isolate until receipt of confirmatory laboratory PCR test result.
3. Ensure confirmatory laboratory-based PCR testing is performed within 24 hours.
4. Report the preliminary positive result to the local Public Health unit as soon as possible.
Do individuals need to provide consent every time they are tested?
The health practitioner administering the COVID-19 test must obtain the consent of the individual in accordance with the Health Care Consent Act, 1996. An individual must consent
to a test for COVID-19 before the test can be administered— this includes staff, student placements, volunteers, caregivers, support workers and general visitors.

 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!