Home Use POCT Oral Fluid Antigen Rapid Test Kit Sputum Saliva Self Test of Covid-19 Corona
Saliva Self Test | Ujian Kendiri Air Liur
Adavantages!!!
Fast: 15 Minutes;
Simple: Easy sample collection;
Cheap: Lower cost with high efficiency;
Stable: Saliva sample is stable at room temperature for convenient transportation;
Flexible: Sample can be easily collected in the laboratory, outpatient clinic, and even at home;
Widely used: Especially workable for children and patients whom cannot easily collect blood sample from.
【MAIN CONTENTS】
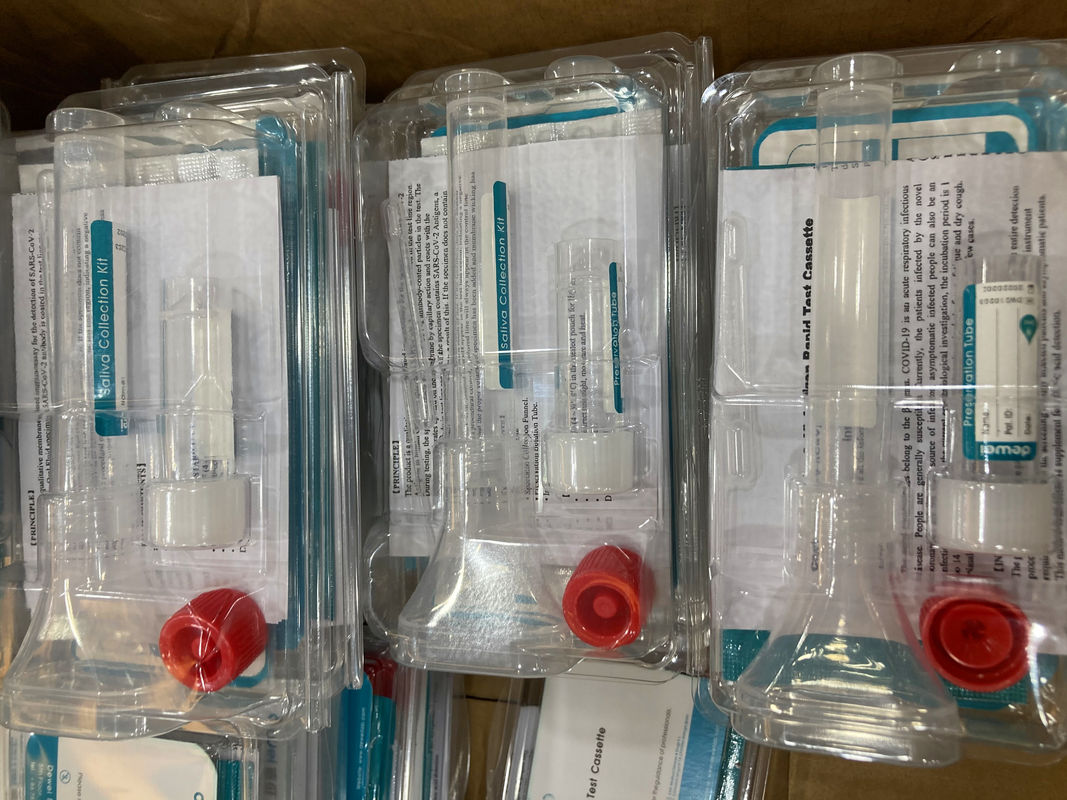
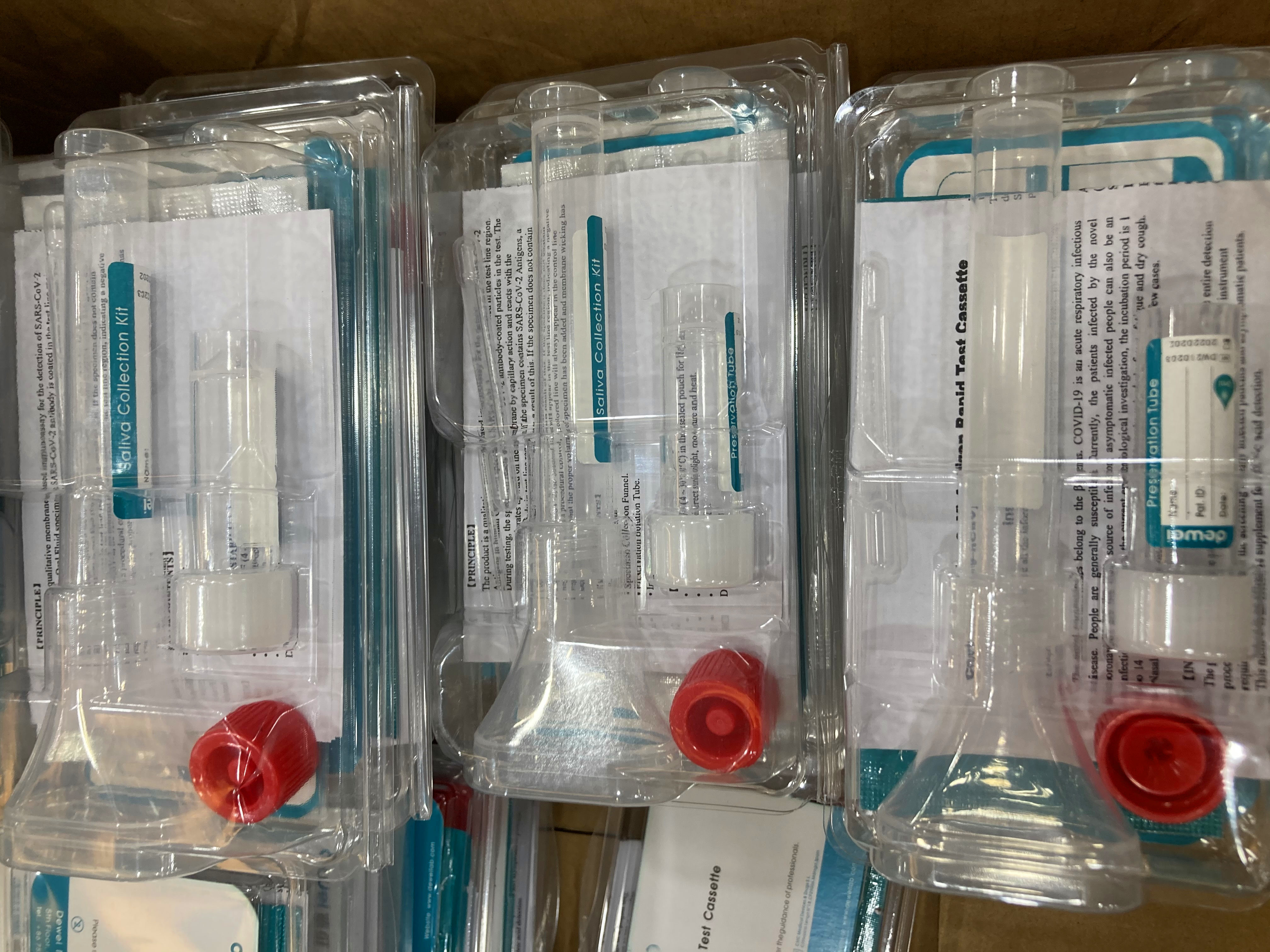
• One pouch containing a reaction test Cassette with desiccant.
• Specimen Collection Kit.
• Instructions for use.
【INTENDED USE】
The product is used for the qualitative detection of SARS-COV-2 infection. The entire detection process takes only 15-20 minutes, and the operation is simple and sensitive. No instrument required. It can be used for the screening of early infected patients and asymptomatic patients. This method is an effective supplement for nucleic acid detection.
【DIRECTION OF USE】
It is recommended to collect the first sputum after deep coughing in the morning.
① Use specimen collection kit to collect oral fluid specimen into preservation solution.
② Gently shake the mixture.
Remove the test cassette from the sealed foil pouch and use it within one hour. Best results will be obtained if the test is performed immediately after opening the foil pouch.
③ Add Extracted specimento the Test Device, Wait for the colored line(s) to appear.
Read the result at 15 minutes. Do not interpret the result after 20 minutes.
1. Who must be tested for COVID-19?
As per the Minister’s Directive: COVID-19: Long-Term Care Home Surveillance Testing and Access to Homes (Minister’s Directive) effective January 8, 2021, all staff, student placement and volunteers working in long-term care homes must be tested regularly in accordance with the Minister’s Directive, unless the exception for individuals who have previously had laboratory confirmed COVID-19 applies. The testing requirements in the Minister’s Directive include all individuals working in long-term care homes who are: • Staff as defined in the Long-Term Care Homes Act, 2007 • Volunteers as defined in the Long-Term Care Homes Act, 2007 • Student placement, meaning any person working in the long-term care home as part of a clinical placement requirement of an educational program of a college or university, and who does not meet the definition of “staff” or “volunteer” under the Long-Term Care Homes Act, 2007. The Minister’s Directive also includes additional testing and documentation requirements for general visitors, caregivers and support workers.
2. When should you perform Dewei COVID-19 Ag Rapid Test?
The COVID-19 Ag Rapid test should NOT be used for diagnosis of COVID-19 infection. Any individual who is symptomatic or a contact of a confirmed case should be directed to their healthcare provider, an assessment centre, or participating licensed community lab to seek PCR testing.
3. If an individual previously tested positive for COVID-19, should they be tested again?
An individual who has previously had laboratory-PCR confirmed COVID-19 and was cleared by the local public health unit (PHU), should generally not be re-tested for surveillance purposes due to persistent shedding. Previously cleared individuals should continue to follow
public health guidance for COVID-19 prevention, including self-isolating after high risk exposures to cases.
Re-testing after clearance should generally only be done with new the onset of new COVID-19 symptoms and can be considered if there is exposure to a confirmed case of COVID-19 or in an outbreak and/or at the direction of the local PHU. Individuals who have previously been
infected with and recovered from COVID-19 should not undergo antigen testing.
4. Do individuals who test positive on the rapid antigen test need to be confirmed with lab-based PCR testing?
A positive test result on the rapid antigen test should be considered a preliminary positive and requires a confirmatory laboratory-based PCR test. The following actions should be taken:
1. Counsel individual that the result is preliminary positive and PCR confirmation is required.
2. Issue guidance to return home and self-isolate until receipt of confirmatory laboratory PCR test result.
3. Ensure confirmatory laboratory-based PCR testing is performed within 24 hours.
4. Report the preliminary positive result to the local Public Health unit as soon as possible.
Please note that Oral collection sensitivity is not as good as nasal swab sample collection.

Please scan here of Operation Video on Youtube:



 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!