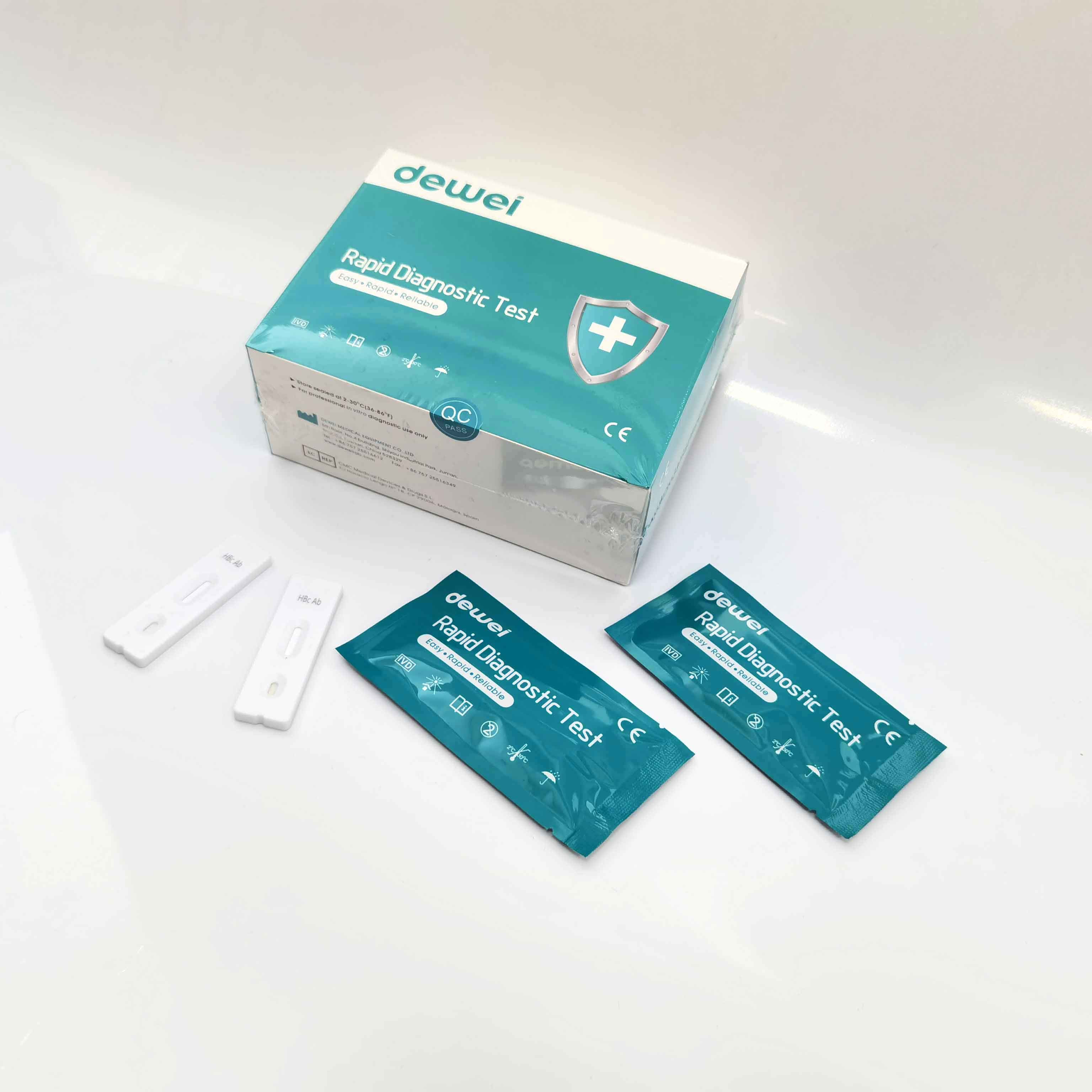
HBcAb Hepatitis B Core Antibody Rapid Detection Kit
【HBcAb INTRODUCTION】
The hepatitis B core antibody (HBcAb) is produced by your immune system after infection by the hepatitis B virus, and it can persist for life. It is a sign that you either have an new, active (acute) hepatitis B infection or that you acquired hepatitis B in the past.
HBcAb is an immune system response to a protein in the core of the virus, and it is only present if you have been infected, rather than immunized against the virus. It is part of a routine screening panel of tests for hepatitis B.
| Test Parameter |
Brand |
Format |
Shelf Life |
Test Time |
| HBcAb |
Dewei |
Cassette |
24 months |
15 mins |
【RAPID TEST INTENDED USE】
HBcAb Rapid Test is a chromatographic immunoassay for the qualitative detection of hepatitis B Core Antibody(HBcAb) in human serum, /plasma.
【DIAGNOSTIC ADVANTAGES】
Results available at 15 mins
Easy to use, no need extra instruments
Results can be read visually
Cost-effective test kit for the detection of Hepatitis B core antibodies
【RAPID TEST SPECIMEN】
Serum/Plasma
【MAIN CONTENTS】
Individually sealed foil pouches containing a test cassette with desiccant
Disposable pipettes
Buffer
Insert
【HBcAb TESTING MANUAL】
Bring tests, specimens, and/or controls toroomtemperature (15-30°C) before use.
1. Remove the test from its sealed pouch, andplaceitonaclean, level surface. Label the cassette with patient orcontrolidentification. For best results, the assay shouldbeperformedwithin one hour.
2. Using the provided disposable pipette, transfer 2dropsofserum or plasma specimen to the specimen well (S)ofthecassette and start the timer.
OR Transfer 1 drop of whole blood specimen to the specimenwell(S) of the cassette with the provided disposablepipette,thenadd 1 drop of buffer and start the timer.
Avoid trapping air bubbles in the specimenwell (S), anddonot add any solution to the result window. As the test begins to work, color will migrateacrossthemembrane.
3. Wait for the colored band(s) to appear. The result shouldberead at 15 minutes. Do not interpret the result after 20minutes.
【HBcAb RESULT INTERPRETATION】
NEGATIVE RESULT:
If only the C line develops, the result is negative or non-reactive.
POSITIVE RESULT:
If both the C and the T lines develop, the result is positive or reactive.
INVALID:
If no C line develops, the assay is invalid regardless of any color development on the T line as indicated below. Repeat the assay with a new device.
【STORAGE AND STABILITY】
The kit should be stored at 2-30°C until the expiry date is printed on the sealed pouch.
The test must remain in the sealed pouch until use.
Keep away from direct sunlight, moisture and heat. Do not freeze.
Care should be taken to protect the components of the kit from contamination. Do not use it if there is evidence of microbial contamination or precipitation. Biological contamination of dispensing equipment, containers, or reagents can lead to false results.


 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!