cTnI Rapid Test Cassette Troponin I Rapid Test Kit
【cTnI INTRODUCTION】
The creatine troponin complex, consisting of cardiac muscle troponin I (cTnI), troponin T (TnT) and troponin C (TnC), can adjust the interaction between actin and myosin. The concentration of cTnI is typically low in healthy human blood. However, when an acute myocardium infarction occurs, cTnI is released into the blood quickly, which leads to a rapid increase of cTnI concentration within hours and can last 6 to 10 days.
Therefore, the concentration of cTnI in blood or plasma is regarded as a marker for myocardial infarction. The cTnI CLIA Test is intended to detect cTnI in human serum, plasma or whole blood.
【RAPID TEST INTENDED USE】
cTnI Troponin I Test Kit is used for the qualitative determination of cardiac troponin I in blood, plasma or serum. This test is used to evaluate acute myocardial infarction through the measurement of troponin I.
【DIAGNOSTIC ADVANTAGES】
Results available in only 15 minutes
Easy to use, no need extra instruments
Results can be read visually
Cost-effective test kit for the detection of AMI(acute myocardium infarction)
【RAPID TEST SPECIMEN】
Whole Blood/Serum/Plasma
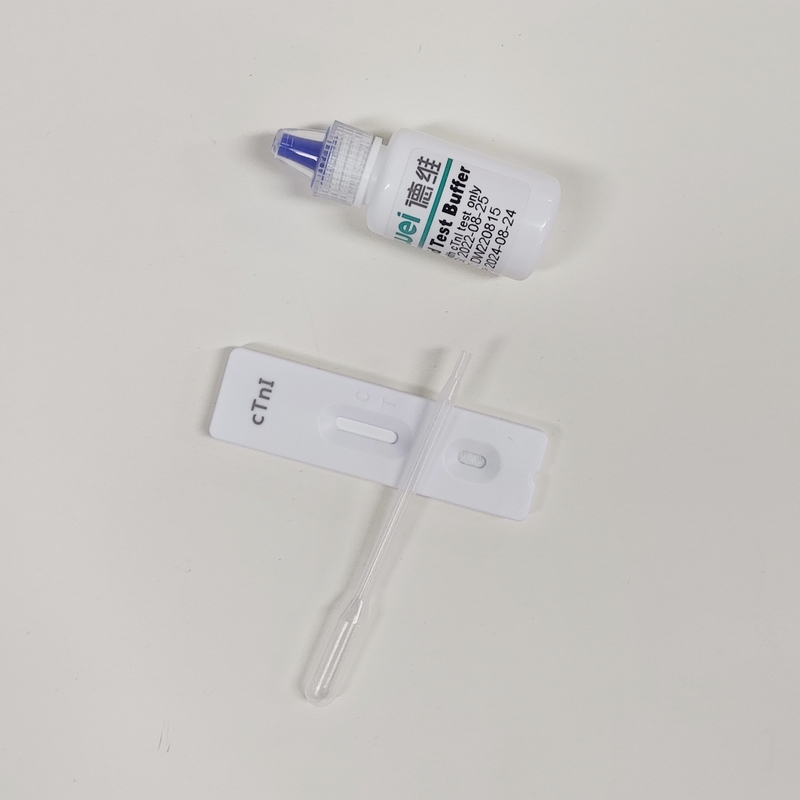
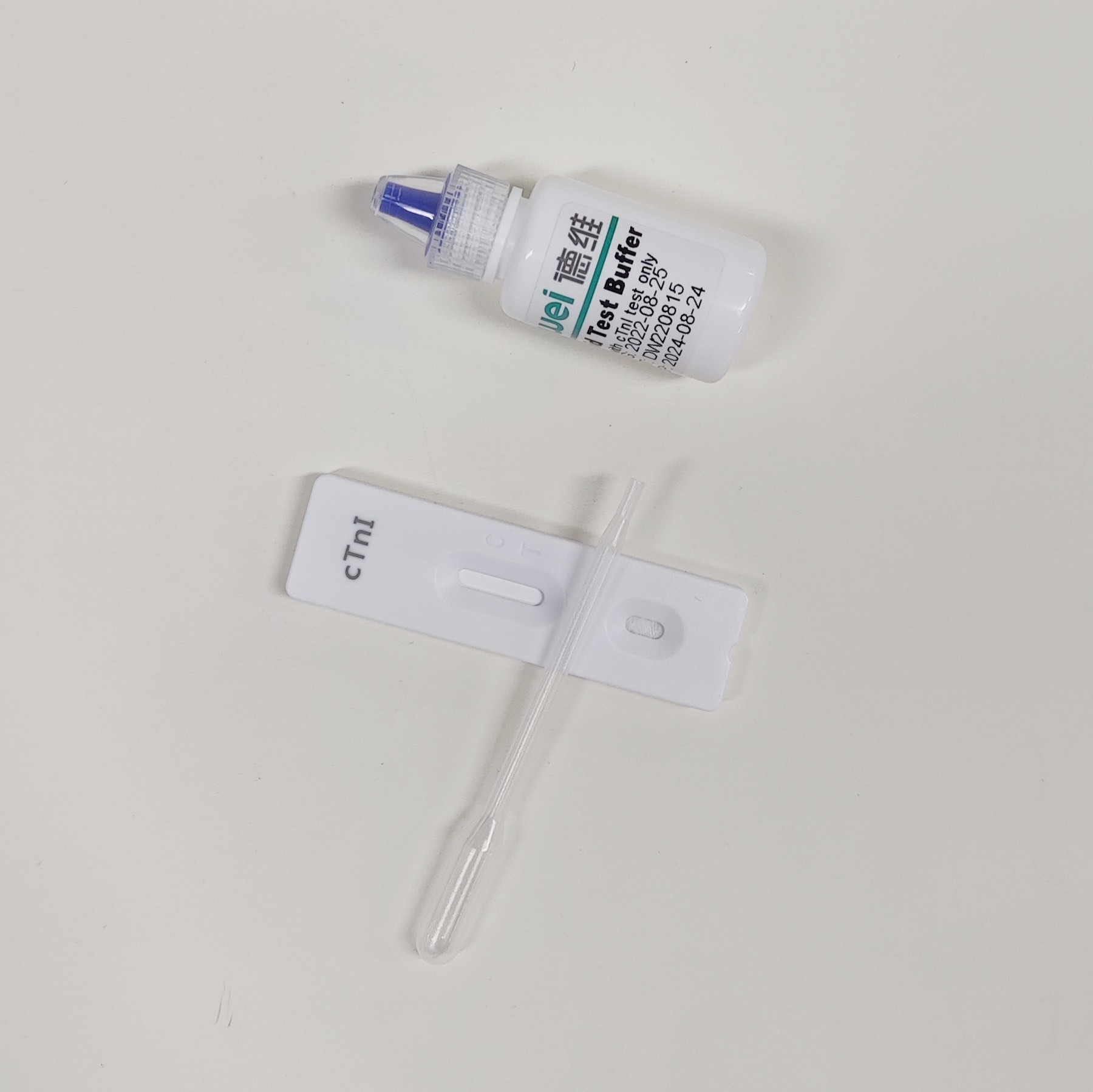
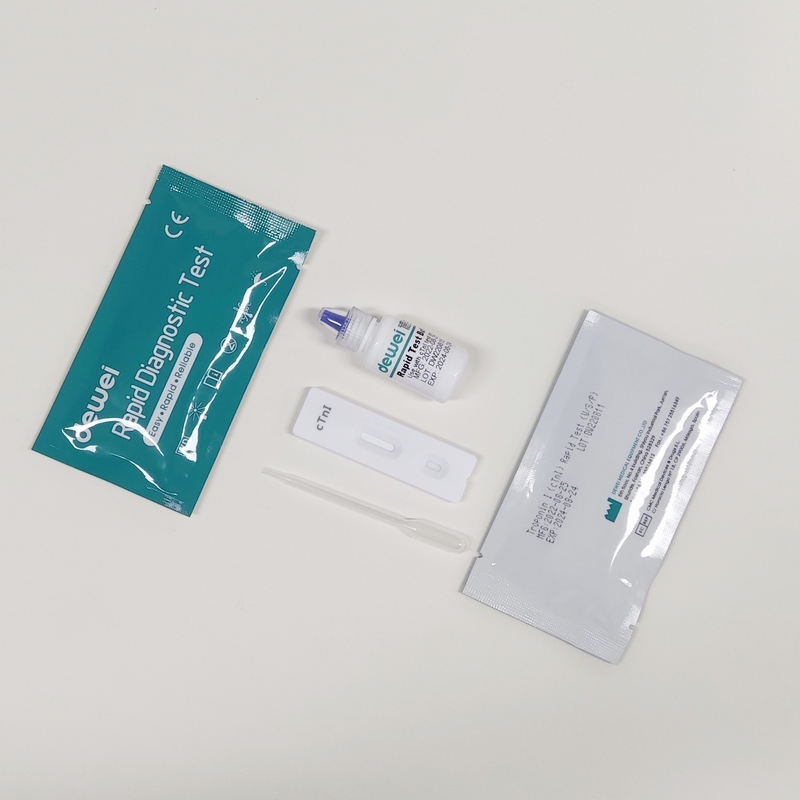
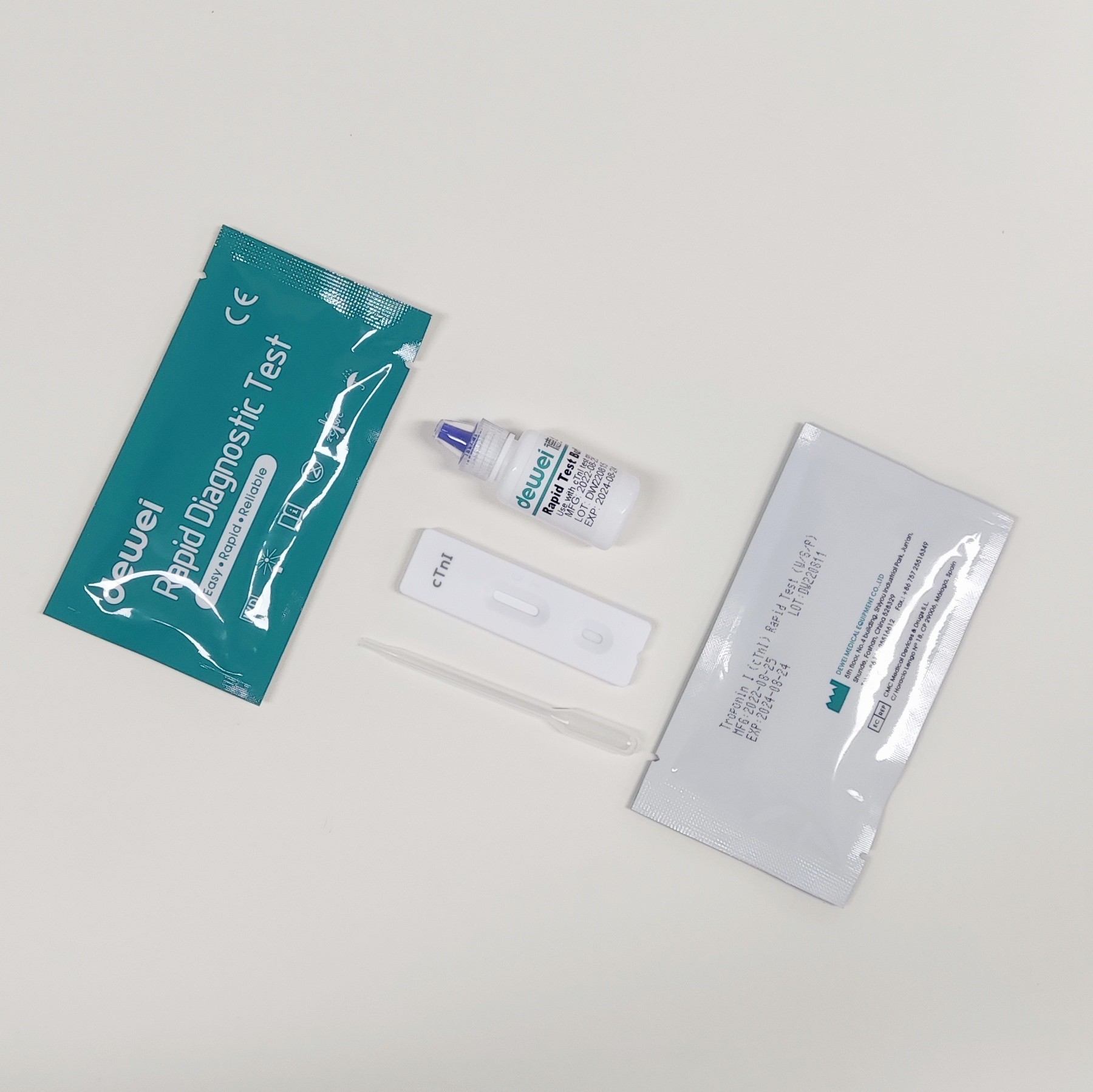
【MAIN CONTENTS】
Individually sealed foil pouches containing a test cassette with desiccant
Disposable pipettes
Buffer
Insert
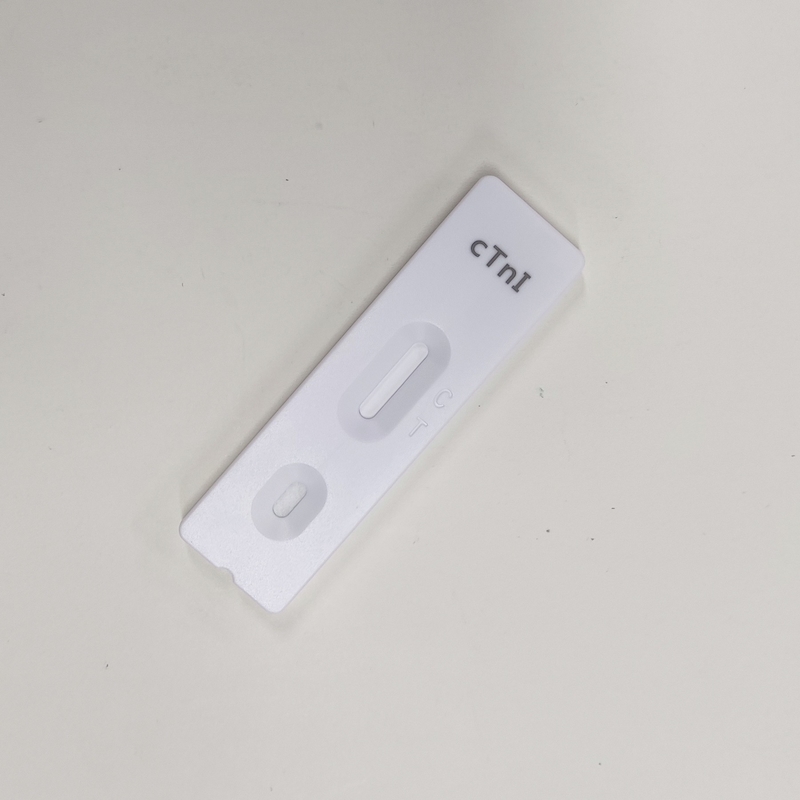
【cTnl RESULT INTERPRETATION】
NEGATIVE RESULT:
If only the C line develops, the result is negative or non-reactive.
POSITIVE RESULT:
If both the C and the T lines develop, the result is positive or reactive.
INVALID:
If no C line develops, the assay is invalid regardless of any color development on the T line as indicated below. Repeat the assay with a new device.
【STORAGE AND STABILITY】
The kit should be stored at 2-30°C until the expiry date is printed on the sealed pouch.
The test must remain in the sealed pouch until use.
Keep away from direct sunlight, moisture and heat. Do not freeze.
Care should be taken to protect the components of the kit from contamination. Do not use it if there is evidence of microbial contamination or precipitation. Biological contamination of dispensing equipment, containers, or reagents can lead to false results.


 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!