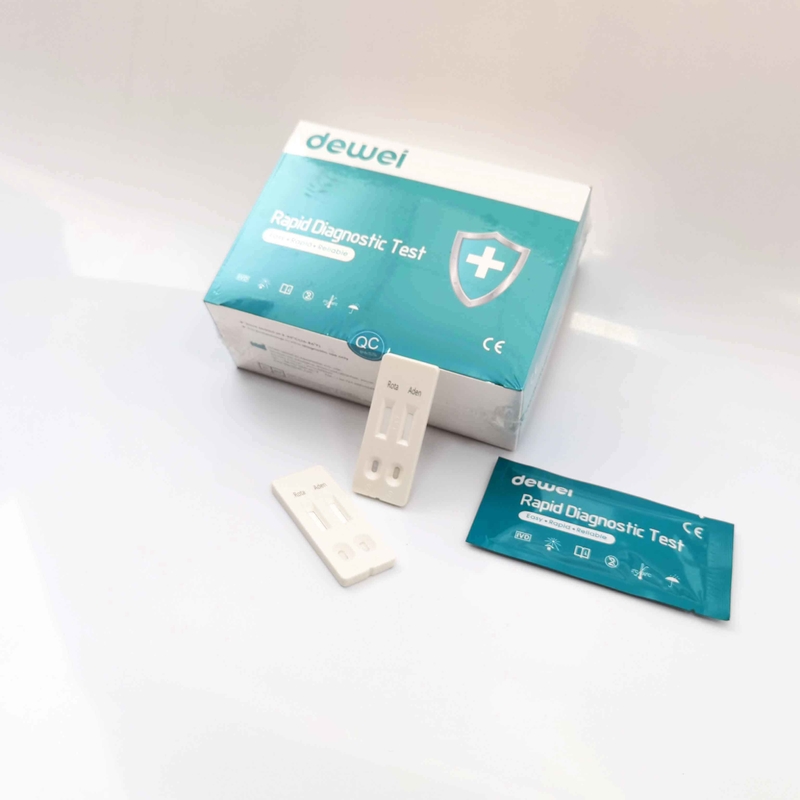
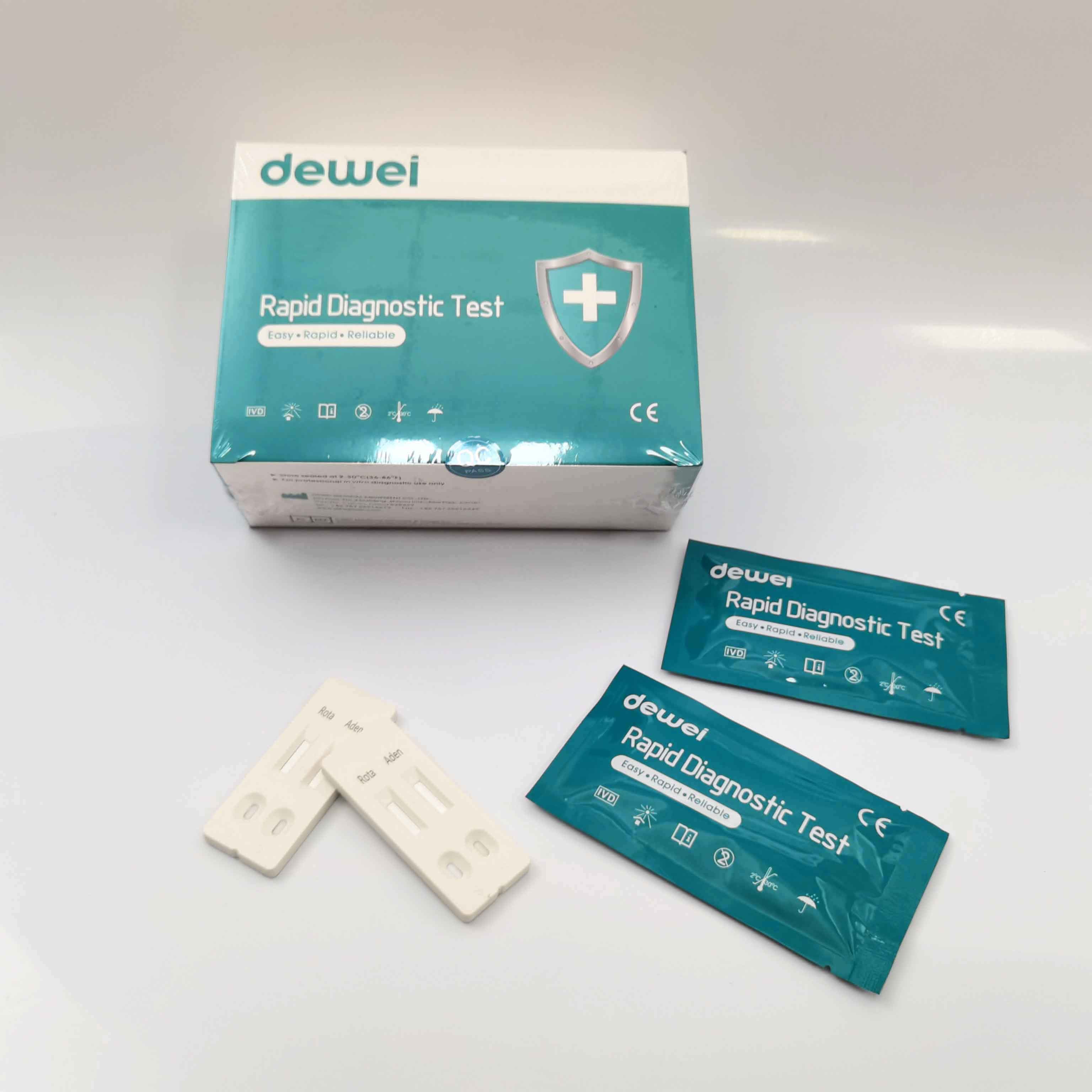
Rotavirus and Adenovirus Combo Rapid Diagnostic Test Device Strip Cassette For Feces Stool
INTENDED USE
The Rotavirus and Adenovirus Combo Rapid Test (Feces) is a rapid visual immunoassay for the qualitative presumptive detection of rotavirus and adenovirus in human fecal specimens. This kit is intended to be used as an aid in the diagnosis of rotavirus and adenovirus infection.
INTRODUCTION
Rotavirus is the most common agent responsible for acute gastroenteritis, mainly in young children. Its discovery in 1973 and its association with infantile gastro-enteritis represented a very important advancement in the study of gastro-enteritis not caused by acute bacterial infection. Rotavirus is transmitted by oro-faecal route with an incubation period of 1-3 days. Although specimen collections taken within the second and fifth day of the illness are ideal for antigen detection, the rotavirus may still be found while diarrhoea continues. Rotaviral gastroenteritis may result in mortality for populations at risk such as infants, the elderly, and immunocompromised patients. In temperate climates, rotavirus infections occur mainly in the winter months. Endemics as well as epidemics affecting some thousand people have been reported. With hospitalised children suffering from acute entric disease up to 50% of the analysed specimen were positive for rotavirus. The viruses replicate in the cell nucleus and tend to be host species specific producing a characteristic cytopathic effect (CPE). Because rotavirus is extremely difficult to culture, it is unusual to use isolation of the virus in diagnosing an infection. Instead, a variety of techniques have been developed to detect rotavirus in feces.
Acute diarrheal disease in young children is a major cause of morbidity worldwide and is a leading cause of mortality in developing countries. Research has shown that enteric adenoviruses, primarily Ad40 and Ad41, are a leading cause of diarrhea in many of these children, second only to the rotaviruses. These viral pathogens have been isolated throughout the world, and can cause diarrhea in children year round. Infections are most frequently seen in children less than two years of age, but have been found in patients of all ages. Further studies indicate that adenoviruses are associated with 4-15% of all hospitalized cases of viral gastroenteritis.
Rapid and accurate diagnosis of gastroenteritis due to adenovirus is helpful in establishing the etiology of gastroenteritis and related patient management. Other diagnostic techniques such as electron microscopy (EM) and nucleic acid hybridization are expensive and labor-intensive. With the self-limiting nature of adenovirus infection, such expensive and labor-intensive tests may not be necessary.
PRINCIPLE
The Rotavirus and Adenovirus Combo Rapid Test (Feces) has been designed to detect rotavirus and adenovirus through visual interpretation of color development in the internal strip. The membrane was immobilized with anti-rotavirus antibodies and anti-adenovirus on the test region. During the test, the specimen is allowed to react with colored anti-rotavirus antibodies colloidal gold conjugates and anti-adnovirus antibodies colloidal gold conjugates, which were precoated on the sample pad of the test. The mixture then moves on the membrane by a capillary action, and interact with reagents on the membrane. If there were enough rotavirus in specimens, a colored band will form at the R region of the membrane. Similarily, If there were enough adenovirus in specimens, a colored band will form at the A region of the membrane. Presence of colored band(s) indicates a positive result, while its absence indicates a negative result. Appearance of a colored band at the control region serves as a procedural control. This indicates that proper volume of specimen has been added and membrane wicking has occurred.
MAIN CONTENTS
• One pouch containing a reaction test Cassette with desiccant.
• Disposable pipettes.
• Buffer.
• Instructions for use.
STORAGE AND STABILITY
• Store at 39~ 86 º F (4 ~ 30 º C) in the sealed pouch for 18 months.
PRECAUTIONS
• For in vitro diagnostic use only.
• Do not use after expiration date.
• The test Cassette should remain in the sealed pouch until use.
• The used test Cassette should be discarded according to local regulations.
SPECIMEN COLLECTION AND STORAGE
The Rotavirus and Adnovirus Combo Rapid Test (Feces) is intended only for use with human fecal specimens.Viral detection is improved by collecting the specimens at the onset of the symptoms. It has been reported that the maximum excretion of rotavirus in the feces of patients with gastroenteritis occurs 3-5 days after onset of symptoms. If the specimens are collected long after the onset of diarrheic symptoms, the quantity of antigen may not be sufficient to obtain a positive reaction or the antigens detected may not be linked to the diarrheic episode.Perform the testing immediately after the specimen collection. Do not leave the specimens at room temperature for prolonged periods. Specimens may be stored at 2-8°C for up to 72 hours.Bring specimens to room temperature prior to testing.Pack the specimens in compliance with applicable regulations for transportation of etiological agents, in case they need to be shipped.
TEST PROCEDURE
*Bring tests, specimens, buffer and/or controls to room temperature (15-30°C) before use.
*Specimen collection and pre-treatment:
*Use the specimens collection cards provided in the kit for specimens collection. Follow the operation procedure written on it for instructions. Other clean dry containers could also be used for the same purpose. Best results will be obtained if the assay is performed within 6 hours after collection.
*For solid specimens: Unscrew and remove the dilution tube applicator. Be careful not to spill or spatter solution from the tube. Collect specimens by inserting the applicator stick into at least 3 different sites of the feces to collect approximately 50 mg of feces (equivalent to 1/4 of a pea).
*For liquid specimens: Hold the pipette vertically, aspirate fecal specimens, and then transfer 2 drops (approximately 50 µL) into the specimen collection tube containing the extraction buffer.
*Place the applicator back into the tube and screw the cap tightly. Be careful not to break the tip of the dilution tube.
*Shake the specimen collection tube vigorously to mix the specimen and the extraction buffer. Specimens prepared in the specimen collection tube may be stored for 6 months at -20°C if not tested within 1 hour after preparation.
*Testing
*Remove the test from its sealed pouch, and place it on a clean, level surface. Label the test with patient or control identification. To obtain a best result, the assay should be performed within one hour.
*Using a piece of tissue paper, break the tip of the dilution tube. Hold the tube vertically and dispense 2 drops of solution into the specimen well (S) of the test device.
*Avoid trapping air bubbles in the specimen well (S), and do not drop any solution in observation window.
*As the test begins to work, you will see color move across the membrane.
*Wait for the colored band(s) to appear. The result should be read at 10 minutes. Do not interpret the result after 20 minutes.
*Note: If the specimen does not migrate (presence of particles), centrifuge the extracted specimens contained in the extraction buffer vial. Collect 80 µL of supernatant, dispense into the specimen well (S) of a new test device and start afresh following the instructions mentioned above.
INTERPRETATION
POSITIVE: Two colored bands appear on the membrane. One band appears in the control region (C) and another band appears in the test region (T).
NEGATIVE: Only one colored band appears, in the control region (C). No apparent colored band appears in the test region (T).
INVALID: Control band fails to appear. Results from any test which has not produced a control band at the specified read time must be discarded. Please review the procedure and repeat with a new test. If the problem persists, discontinue using the kit immediately and contact your local distributor.
FRQ:
| Rotavirus |
Answer |
Adenovirus |
Answer |
| 1) What are the first symptoms of rotavirus? |
A rotavirus infection usually starts within two days of exposure to the virus. Early symptoms are a fever and vomiting, followed by three to seven days of watery diarrhea. The infection can cause abdominal pain as well. |
1) What is the cure for adenovirus?
|
Fortunately, most infections are self-limited in the setting of a normal immune response and do not warrant specific therapy. Therapy for most adenovirus infections is supportive and symptomatic, except in the case of immunocompromised individuals who are at risk fo increased morbidity and mortality.
|
| 2) What causes rotavirus in adults? |
Spread by faecal-oral transmission, rotavirus infection in adults typically manifests with nausea, malaise, headache, abdominal cramping, diarrhoea, and fever. Infection can also be symptomless. |
2) What are the symptoms of adenovirus?
|
Adenoviruses most often infect the airways leading to cold-like symptoms, including sore throat, sneezing, runny nose, cough, headache, chills, or symptoms of croup or bronchitis. Some people might also get a fever. |
|
3) How to get rid of rotavirus?
|
There's no specific treatment for a rotavirus infection. Antibiotics and antivirals won't help a rotavirus infection. Usually, the infection resolves within three to seven days. Preventing dehydration is the biggest concern. |
3) How long is contagious adenovirus?
|
Adenovirus infections are most communicable during the first few days of acute illness, but persistent and intermittent shedding of the virus for longer periods is frequent and reinfection can occur. |
|
4) How long can rotavirus last?
|
Signs and symptoms
Symptoms of rotavirus usually start about 2 days after a person is exposed to the virus. Vomiting and watery diarrhea can last 3 to 8 days. |
4) Do you need antibiotics for adenovirus?
|
There is no specific medical treatment for adenovirus infections, and antibiotics will not work on adenoviruses. There is no good evidence that antiviral drugs work with adenovirus infections in children with normal immune systems. |
|
5) What is the common side effect of rotavirus?
|
Rotavirus can be very harmful. Diarrhea, vomiting, and fever can cause a loss of body fluids. This leads to dehydration, which can be very dangerous, especially for babies and young children. Some children need an IV (needle in their vein) in the hospital to replace lost fluids. |
5) Is adenovirus harmful?
|
Most infections are mild, but serious infections can happen, especially in infants and people with weak immune systems. Some types of the virus are linked to more severe disease. |
|
6) Who is at risk for rotavirus?
|
Nearly every child is infected with rotavirus at least once before the age of five, with the first infection usually occurring before age three. Person-to-person transmission through the hands appears to be responsible for the virus spreading in closed environments, such as homes and hospitals. |
6) What organ does adenovirus affect?
|
Adenoviruses are a group of common viruses that can infect your airways and lungs, eyes, intestines, urinary tract, and nervous system. They're common causes of fever, coughs, sore throats, diarrhea, and pink eye. |
|
7) What kills rotavirus?
|
Rotavirus is a tough virus. It can live on objects for several days unless it is killed by a disinfectant (cleaner that kills germs). It is very hard to prevent rotavirus with just hand-washing and cleaning with a disinfectant. Vaccination is the best way to keep children safe from rotavirus. |
7) How do you confirm adenovirus?
|
How are adenovirus infections diagnosed in a child?
Blood tests.
Swabbing eye, nose, or throat for testing.
Stool sample testing.
Chest X-ray.
|
|
8) How to detect rotavirus?
|
The rotavirus antigen test detects rotavirus in the feces. This is the most common cause of infectious diarrhea in children. A stool sample is obtained to test for a rotavirus infection. Rotavirus is the most common cause of infectious diarrhea in children. |
8) Does adenovirus cause cough?
|
The virus most commonly infects your respiratory system. An adenovirus infection in your respiratory tract can cause symptoms similar to the common cold or flu. Symptoms or conditions you may experience include: Cough. |
|
9) What food sources cause rotavirus?
|
Role of foods
Drinking water, water from the public supply and recreational water may be contaminated and involved in the transmission of rotaviruses to humans. All kinds of food can be involved: foods of animal or plant origin, raw or undercooked foods, ready-to-eat foods or food ingredients. |
9) Does adenovirus go away on its own?
|
Most adenoviral infections are mild and go away without treatment.. |
|
10) What color is rotavirus poop?
|
Anyone caring for small children should know the symptoms of rotavirus, including: Frequent, watery diarrhea (often foul-smelling, green, or brown. |
10) Does adenovirus have a vaccine?
|
Adenovirus vaccine comes as two tablets, taken orally (by mouth) at the same time. The tablets should be swallowed whole, not chewed or crushed. Adenovirus vaccine may be given at the same time as other vaccines. |
For details, please contact Dewei staff for Instruction Manual!

 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!