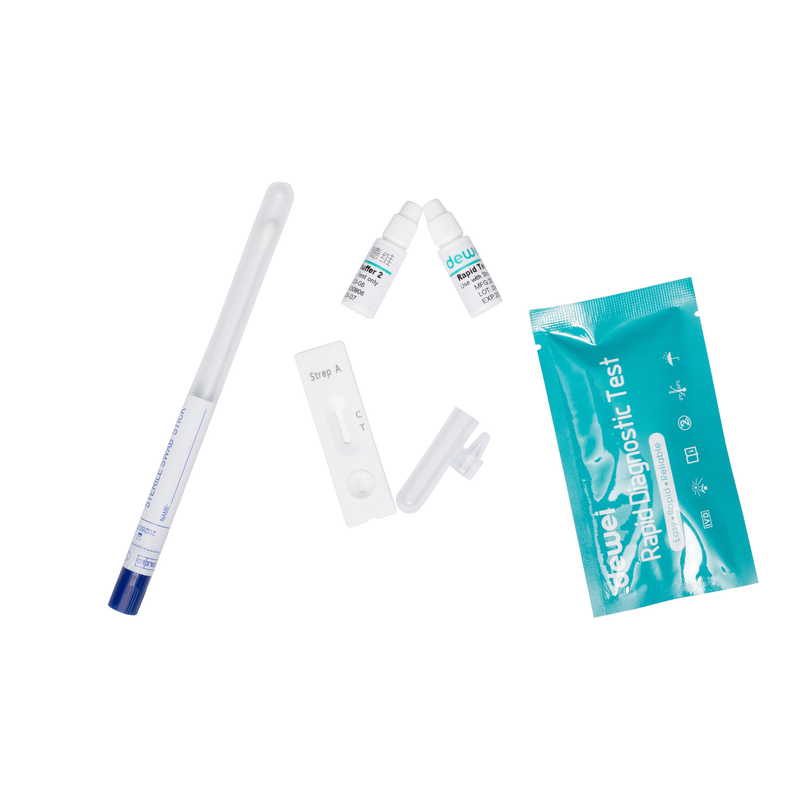
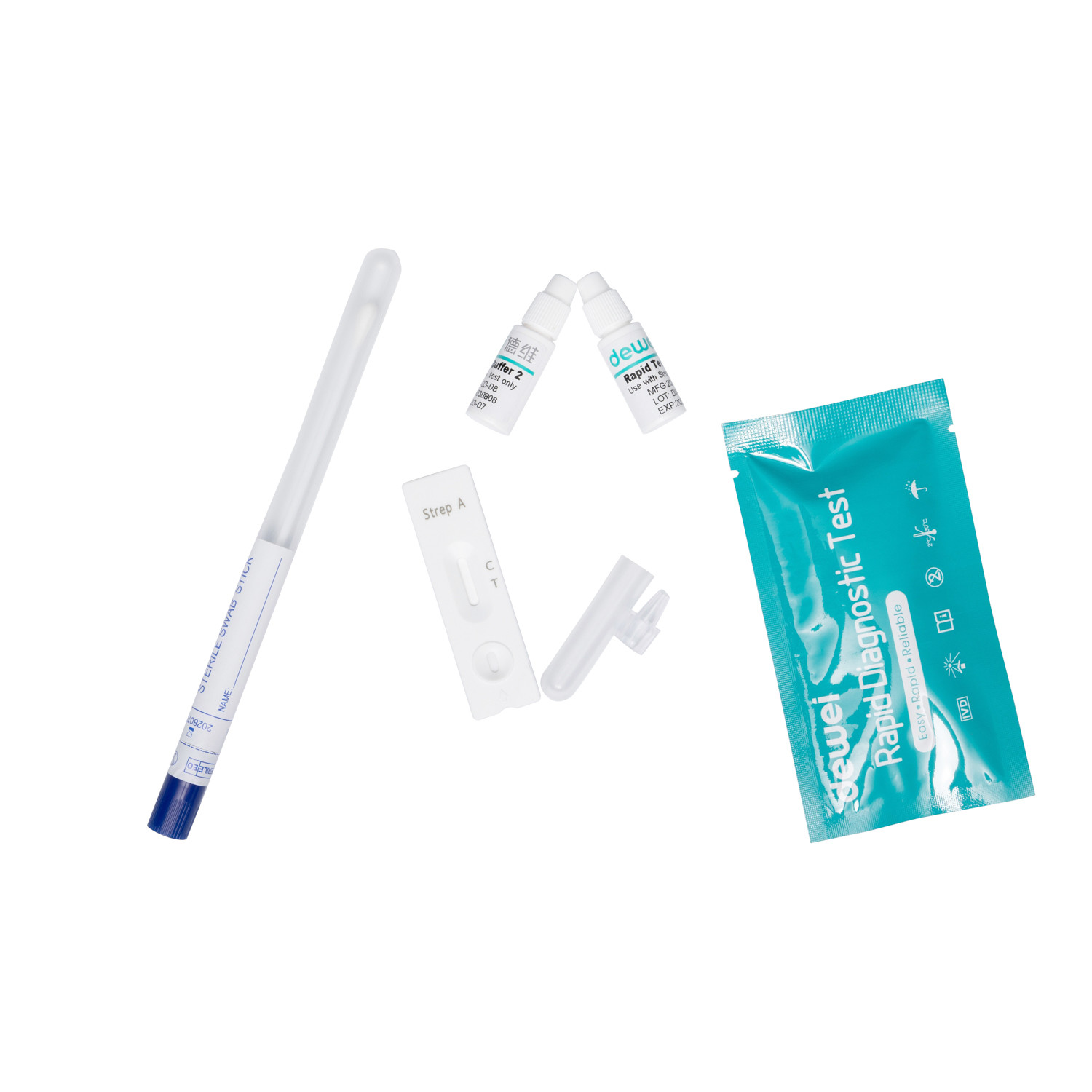
20 25pcs/Box Strep B Rapid Test Kit by Swab Group B Streptococcus (GBS) Antigen
INTENDED USE
The Strep B Rapid Test (Swab) is a rapid visual immunoassay for the qualitative, presumptive detection of Group B Streptococcus (GBS) antigens in specimens taken from vaginal or rectal swaps of pregnant women, or general swabs from newborn. This kit is intended for use as an aid in the diagnosis of Strep B infection.
INTRODUCTION
Group B Streptococci (GBS) or Streptococcus agalactiae are among the most frequent causes of life-threatening infectious in neonates. Between 5% and 30% of all pregnant women are colonized with GBS.1 Several recent studies have shown that the intrapartum treatment of GBS-colonized women significantly reduces the incidence of GBS-caused sepsis. 2-4 The US Center for Disease Control and Prevention (CDC) recommends routine examination for Group B streptococcus between the 35th and the 37th week of pregnancy. A CDC study has shown that a routine examination is 50% more effective than the use of antibiotics for pregnant women with clinical risk factors.
Standard culture methods require 24 to 48 hours, and the results may not be available soon enough for efficient treatment. Thus, methods utilizing more rapid screening techniques are required.
PRINCIPLE
The Strep B Rapid Test (Swab) detects Group B Streptococcus antigens through visual interpretation of color development on the internal strip. Anti-Strep B antibodies are immobilized on the test region of the membrane. During testing, the specimen reacts with polyclonal anti-Strep B antibodies conjugated to colored particles and precoated onto the sample pad of the test. The mixture then migrates through the membrane by capillary action, and interacts with reagents on the membrane. If there is sufficient Strep B antigen in the specimen, a colored band will form at the test region of the membrane. The presence of this colored band indicates a positive result, while its absence indicates a negative result. The appearance of a colored band at the control region serves as a procedural control, indicating that proper volume of specimen has been added and membrane wicking has occurred.
MAIN CONTENTS
- Individually packed test devices
|
Each test contains colored conjugates and reactive reagents precoated at the corresponding regions. |
|
|
For extraction specimen |
|
|
Non-viable Strep B; 0.09% sodium azide |
|
|
For specimen collection |
|
|
For adding specimens |
|
|
For specimen preparation |
|
|
Workstation |
|
|
For operating instructions |
PRECAUTIONS
1. For professional in vitro diagnostic use only.
2. Do not use after the expiration date indicated on the package. Do not use the test if the foil pouch is damaged. Do not reuse tests.
3. This kit contains products of animal origin. Certified knowledge of the origin and/or sanitary state of the animals does not completely guarantee the absence of transmissible pathogenic agents. It is therefore recommended that these products be treated as potentially infectious, and handled by observing usual safety precautions (e.g., do not ingest or inhale).
4. Avoid cross-contamination of specimens by using a new extraction tube for each specimen obtained.
5. Read the entire procedure carefully prior to testing.
6. Do not eat, drink or smoke in any area where specimens and kits are handled. Handle all specimens as if they contain infectious agents. Observe established precautions against microbiological hazards throughout the procedure and follow standard procedures for the proper disposal of specimens. Wear protective clothing such as laboratory coats, disposable gloves and eye protection when specimens are assayed.
7. Do not interchange or mix reagents from different lots. Do not mix solution bottle caps.
8. Use only dacron or rayon tipped sterile swabs with plastic shafts such as those provided. Do not use calcium alginate, cotton tipped, or wooden shafted swabs.
9. Reagents A & B are slightly caustic. Avoid contact with eyes or mucous membranes. In the event of accidental contact, wash thoroughly with water.
10. The positive control contains sodium azide, which may react with lead or copper plumbing to form potentially explosive metal azides. 11. When disposing of these solutions always flush with copious amounts of water to prevent azide buildup.
12. Humidity and temperature can adversely affect results.
13. Used testing materials should be discarded according to local regulations.
SRORAGE
1. The kit should be stored at 2-30°C until the expiry date printed on the sealed pouch.
2. The test must remain in the sealed pouch until use.
3. Do not freeze.
4. Care should be taken to protect components in this kit from contamination. Do not use if there is evidence of microbial contamination or precipitation. Biological contamination of dispensing equipment, containers or reagents can lead to false results.
SPECIMEN COLLECTION AND STORAGE
1. The quality of specimen obtained is of extreme importance. Collect swab specimens using standard clinical methods.
2. Use only Dacron or Rayon tipped sterile swabs with plastic shafts such as those provided. Do not use calcium alginate, cotton tipped, or wooden shafted swabs.
3. It is recommended that swabs specimens be processed as soon as possible after collection. If swabs are not processed immediately, they should be placed into a sterile, dry, tightly capped tube or bottle and refrigerated. Do not freeze. Swabs can be stored at room temperature up to 4 hours, or refrigerated (2-8°C) up to 24 hours. All specimens should be allowed to reach room temperature (15-30°C) before testing.
4. If a liquid transport method is desired, use Modified Stuart’s Transport Media and follow the manufacturer’s instructions. Do not place the swab in any transport device containing medium. Transport medium interferes with the assay, and viability of organisms is not required for the assay. Do not use transport media formulas that include charcoal or agar.
5. If a bacteria culture is desired, lightly roll the swab on a appropriate cell culture plate before using it in the test. The extraction reagents in the test will kill bacteria on swabs and make them impossible to culture.
OPERATION PROCEDURE
Bring tests, specimens, reagents and/or controls to room temperature (15-30°C) before use if the specimens are refrigerated.
1. Prepare swab specimens:
• Place a clean extraction tube in the designated area of the workstation. Add 14 drops of Extraction Buffer to the extraction tube.
• Immediately immerse the swab into the extraction tube. Use a circular motion to roll the swab against the side of the extraction tube so that the liquid is expressed from the swab and can reabsorb.
• Let stand for 1 minute at room temperature, and then squeeze the swab firmly against the tube to expel as much liquid as possible from the swab. Cap the extraction tube with the attached dropper tip. Discard the swab following guidelines for handling infectious agents.
2. Remove the test from its sealed pouch, and place it on a clean, level surface. Label the device with patient or control identification. For best results, the assay should be performed within one hour.
3. Add 3 drops (approximately 120 µL) of extracted solution from the extraction tube to the sample well on the test device.
Avoid trapping air bubbles in the specimen well (S), and do not add any solution to the observation window.
As the test begins to work, color will migrate across the membrane.
4. Wait for the colored band(s) to appear. The result should be read at 10 minutes. Do not interpret the result after 15 minutes.
INTERPRETATION
POSITIVE: Two colored bands appear on the membrane. One band appears in the control region (C) and another band appears in the test region (T).
NEGATIVE: Only one colored band appears, in the control region (C). No apparent colored band appears in the test region (T).
INVALID: Control band fails to appear. Results from any test which has not produced a control band at the specified read time must be discarded. Please review the procedure and repeat with a new test. If the problem persists, discontinue using the kit immediately and contact your local distributor.
FRQ:
1) How did I get strep B?
Group B Strep is not a sexually transmitted disease. Like many bacteria, GBS can be passed from one person to another through direct contact, for example, hand holding, kissing, close physical contact, etc. As GBS is often found in the vaginaand rectum, it can also be passed on during sex.
2) Does strep B ever go away?
Remember, group B strep is a normal bacteria that can come and go throughout your life. Yes, it might go away, but it can also come back. It's important to know that testing positive means nothing for your overall health, and there's no need to worry about it after pregnancy.
3) Is strep B very contagious?
Group B Strep is not a sexually transmitted disease. Like many bacteria, GBS may be passed from one person to another through direct contact, for example, hand-holding, kissing, close physical contact, etc. As GBS bacteria are often found in the vaginaand rectum, they can also be passed on during sex.
4) What happens if you test positive for group B strep?
Doctors will test a pregnant woman to see if she has GBS. If she does, she will get intravenous (IV) antibiotics during labor to kill the bacteria. Doctors usually use penicillin, but can give other medicines if a woman is allergic to it. It's best for a woman to get antibiotics for at least 4 hours before delivery.
5) Is GBS a STD or STI?
Group B strep colonization is not a sexually transmitted disease (STD).. One of every four or five pregnant women carries GBS in the rectum orvagina. A fetus may come in contact with GBS before or during birth if the mother carries GBS in the rectum orvagina.
6) Can a man carry strep B?
Group B strep is a type of bacteria called streptococcal bacteria. It's very common in both men and women and usually lives in the bottom (rectum) orvagina. Group B strep is normally harmless and most people will not realise they have it.
Can I pass group B strep to my husband?
7) Group B strep bacteria aren't sexually transmitted, and they're not spread through food or water. How the bacteria are spread to anyone other than newborns isn't known. Group B strep can spread to a baby during a vaginal delivery if the baby is exposed to — or swallows — fluids containing group B strep.
8) What does strep B discharge look like?
Even though health care providers do not widely recognize GBS vaginitis, GBS can cause yellow or green discharge as well as vaginal burning and/or irritation. These symptoms may be mistaken for a yeast infection or bacterial vaginosis.
9) What antibiotic kills strep B?
GBS infections are usually treated with penicillin. Sometimes other types of antibiotics are used. Soft tissue and bone infections may require surgery to treat.
10) Which is worse, strep A or B?
Strep A infections are found on the surface of the skin and inside the throat, causing a number of health problems such as strep throat and scarlet fever. Strep B, meanwhile, lives harmlessly in the digestive system and in thevagina.




 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!