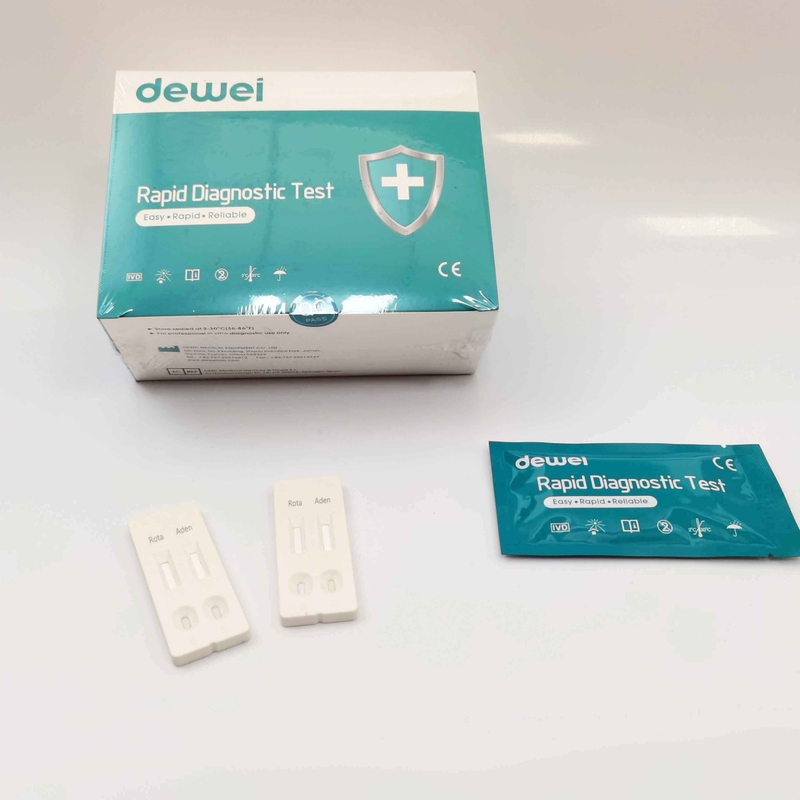
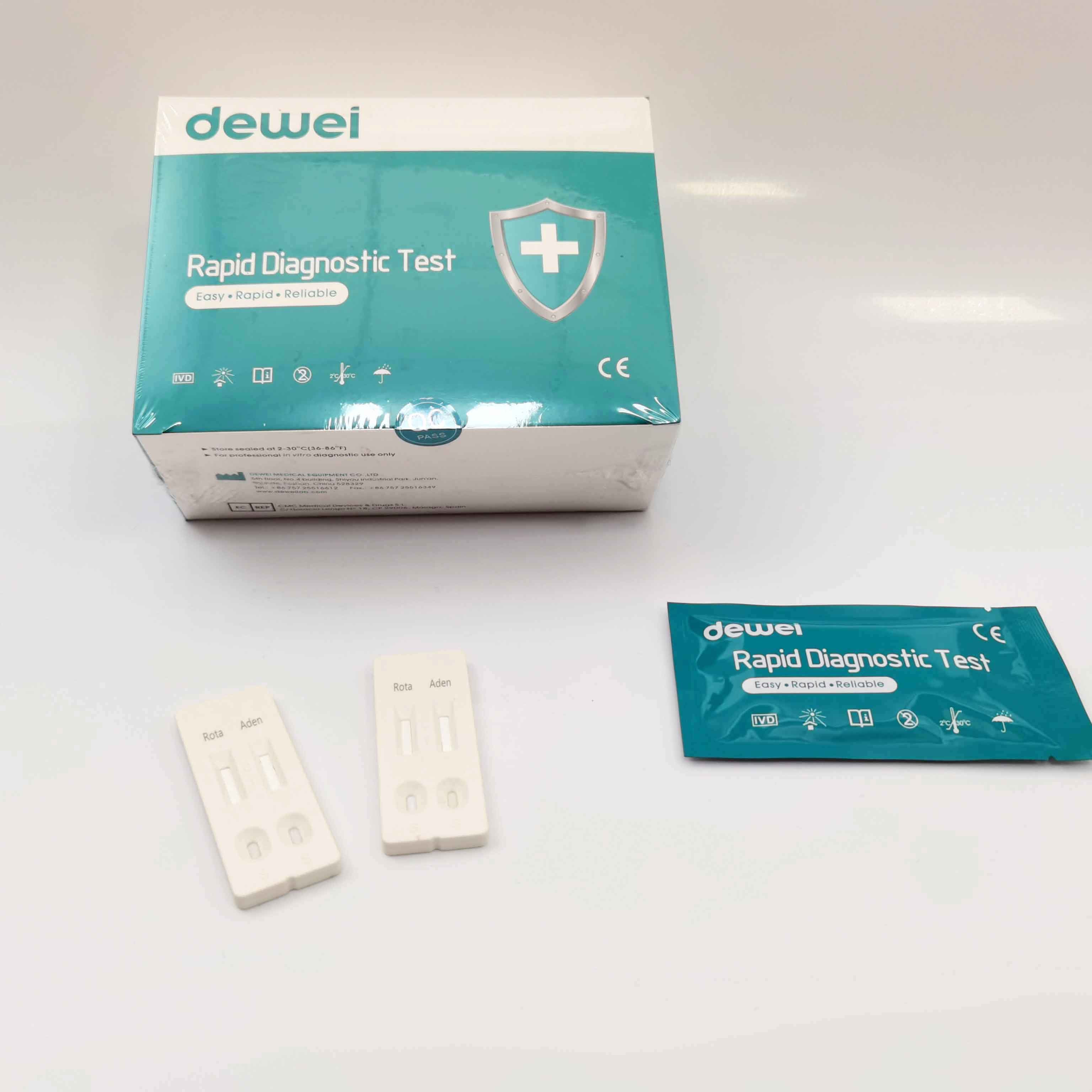
1) What are the first symptoms of rotavirus?
A rotavirus infection usually starts within two days of exposure to the virus. Early symptoms are a fever and vomiting, followed by three to seven days of watery diarrhea. The infection can cause abdominal pain as well.
2) What causes rotavirus in adults?
Spread by faecal-oral transmission, rotavirus infection in adults typically manifests with nausea, malaise, headache, abdominal cramping, diarrhoea, and fever. Infection can also be symptomless.
3) How to get rid of rotavirus?
There's no specific treatment for a rotavirus infection. Antibiotics and antivirals won't help a rotavirus infection. Usually, the infection resolves within three to seven days. Preventing dehydration is the biggest concern.
4) How long can rotavirus last?
Signs and symptoms
Symptoms of rotavirus usually start about 2 days after a person is exposed to the virus. Vomiting and watery diarrhea can last 3 to 8 days.
5) What is the common side effect of rotavirus?
Rotavirus can be very harmful. Diarrhea, vomiting, and fever can cause a loss of body fluids. This leads to dehydration, which can be very dangerous, especially for babies and young children. Some children need an IV (needle in their vein) in the hospital to replace lost fluids.
6) Who is at risk for rotavirus?
Nearly every child is infected with rotavirus at least once before the age of five, with the first infection usually occurring before age three. Person-to-person transmission through the hands appears to be responsible for the virus spreading in closed environments, such as homes and hospitals.
7) What kills rotavirus?
Rotavirus is a tough virus. It can live on objects for several days unless it is killed by a disinfectant (cleaner that kills germs). It is very hard to prevent rotavirus with just hand-washing and cleaning with a disinfectant. Vaccination is the best way to keep children safe from rotavirus.
8) How to detect rotavirus?
The rotavirus antigen test detects rotavirus in the feces. This is the most common cause of infectious diarrhea in children. A stool sample is obtained to test for a rotavirus infection. Rotavirus is the most common cause of infectious diarrhea in children.
9) What food sources cause rotavirus?
Role of foods
Drinking water, water from the public supply and recreational water may be contaminated and involved in the transmission of rotaviruses to humans. All kinds of food can be involved: foods of animal or plant origin, raw or undercooked foods, ready-to-eat foods or food ingredients.
10) Is milk good for rotavirus?
Milk contains sugars, proteins, and antibodies capable of inhibiting rotaviruses.
11) What color is rotavirus poop?
Anyone caring for small children should know the symptoms of rotavirus, including: Frequent, watery diarrhea (often foul-smelling, green, or brown.
Adenovirus:
1) What is the cure for adenovirus?
Fortunately, most infections are self-limited in the setting of a normal immune response and do not warrant specific therapy. Therapy for most adenovirus infections is supportive and symptomatic, except in the case of immunocompromised individuals who are at risk fo increased morbidity and mortality.
2) What are the symptoms of adenovirus?
Adenoviruses most often infect the airways leading to cold-like symptoms, including sore throat, sneezing, runny nose, cough, headache, chills, or symptoms of croup or bronchitis. Some people might also get a fever.
3) How long is contagious adenovirus?
Adenovirus infections are most communicable during the first few days of acute illness, but persistent and intermittent shedding of the virus for longer periods is frequent and reinfection can occur.
4) Do you need antibiotics for adenovirus?
There is no specific medical treatment for adenovirus infections, and antibiotics will not work on adenoviruses. There is no good evidence that antiviral drugs work with adenovirus infections in children with normal immune systems.
5) Is adenovirus harmful?
Most infections are mild, but serious infections can happen, especially in infants and people with weak immune systems. Some types of the virus are linked to more severe disease.
6) What organ does adenovirus affect?
Adenoviruses are a group of common viruses that can infect your airways and lungs, eyes, intestines, urinary tract, and nervous system. They're common causes of fever, coughs, sore throats, diarrhea, and pink eye.
7) How do you confirm adenovirus?
How are adenovirus infections diagnosed in a child?
Blood tests.
Swabbing eye, nose, or throat for testing.
Stool sample testing.
Chest X-ray.
8) Does adenovirus cause cough?
The virus most commonly infects your respiratory system. An adenovirus infection in your respiratory tract can cause symptoms similar to the common cold or flu. Symptoms or conditions you may experience include: Cough.
9) What is the late phase of adenovirus?
The “late” phase is involved in making gene products that are related to production and assembly of capsid proteins. Virus packaged by transfecting HEK 293 cells with adenoviral-based vectors is capable of infecting human cells.
10) Does adenovirus go away on its own?
Most adenoviral infections are mild and go away without treatment.
11) Does adenovirus have a vaccine?
Adenovirus vaccine comes as two tablets, taken orally (by mouth) at the same time. The tablets should be swallowed whole, not chewed or crushed. Adenovirus vaccine may be given at the same time as other vaccines.
12) What is the best treatment for adenovirus?
Like most other viral infections, there are no specific drugs that can target the adenovirus. The body's immune system fights the infection and it resolves in three to five days in most individuals. Bed rest and isolation to prevent spread of infection. Drinking plenty of water and clear fluids.
13) What is the drug of choice for adenovirus?
Medication Summary
Ribavirin and cidofovir therapy have been used with variable success in immunosuppressed hosts. Anecdotal evidence suggests success against adenoviral infection with combined intravenous ribavirin and pooled human intravenous immunoglobulin.
What are the benefits of adenovirus?
14) Adenoviruses are commonly used for gene transfer, as they can be generated at high titers and efficiently infect and express their genes in a variety of cell types including both dividing and quiescent cells.
15) What is the new treatment for adenovirus?
Preliminary results indicate that brincidofovir is safe and very effective at decreasing adenovirus viremia and adenovirus-induced pathogenicity and mortality. Anti-adenovirus adoptive T cell therapy is another very promising approach to treating allo-HSCT patients as demonstrated in clinical studies.

 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!