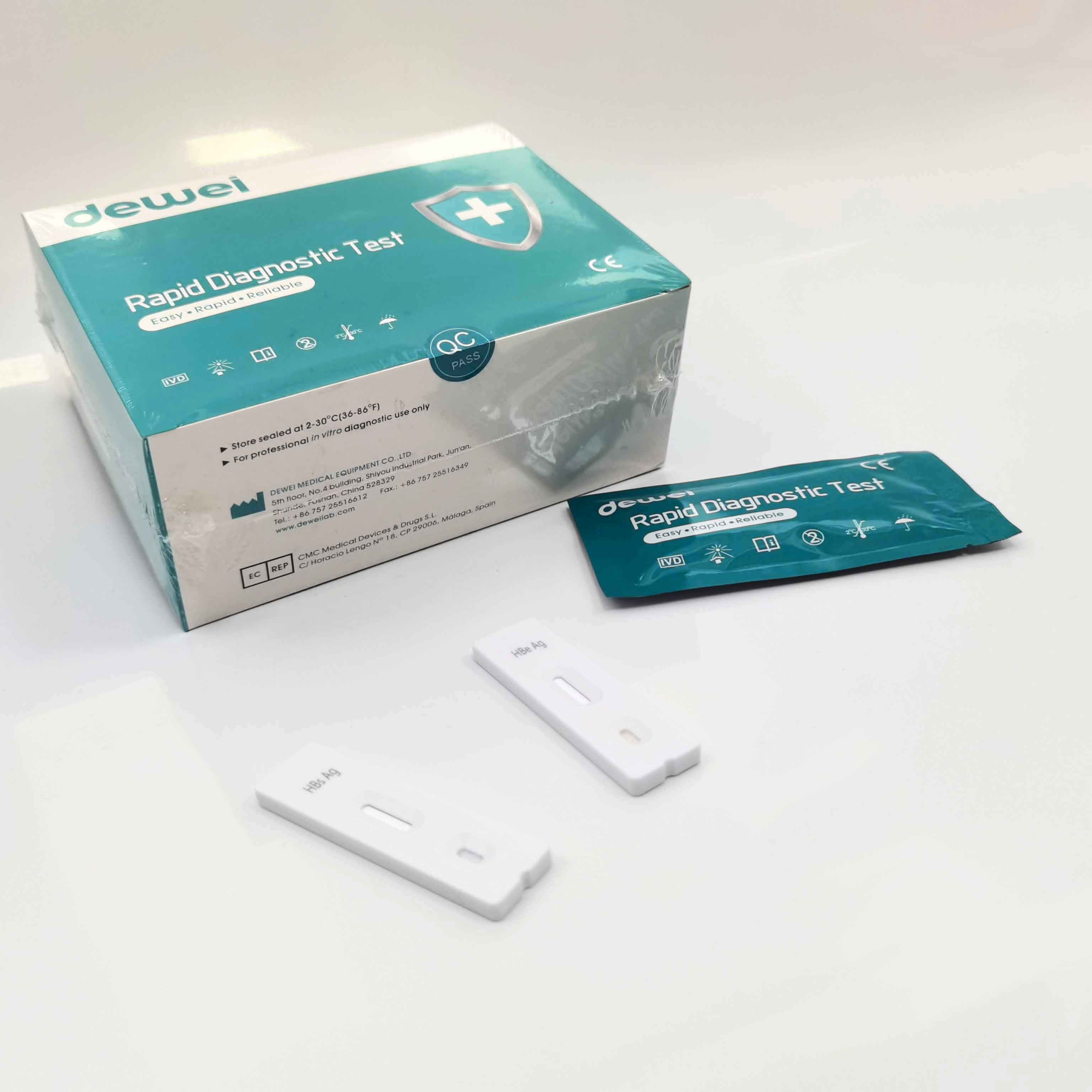
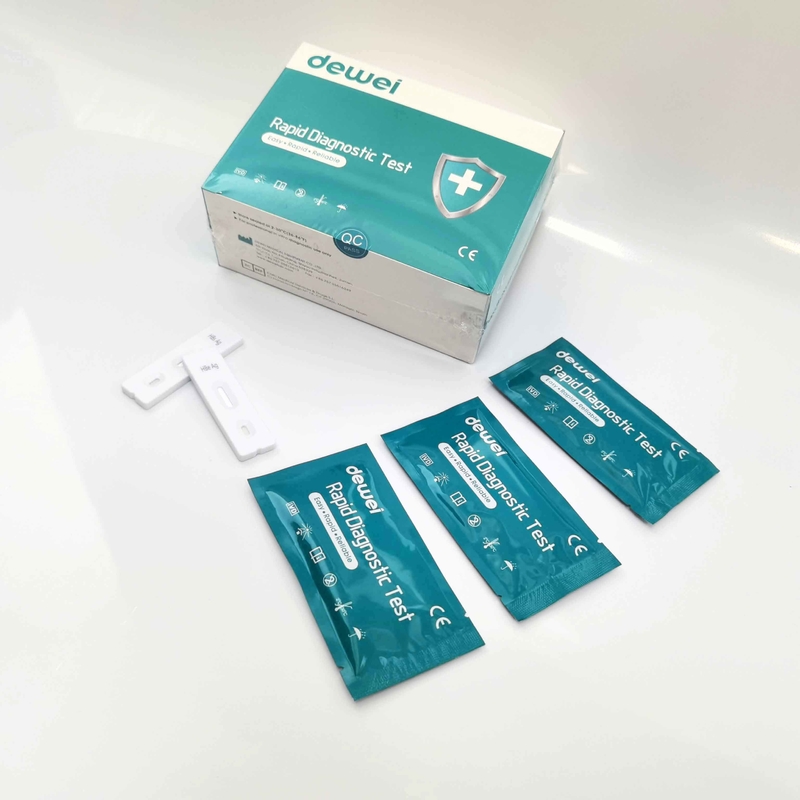
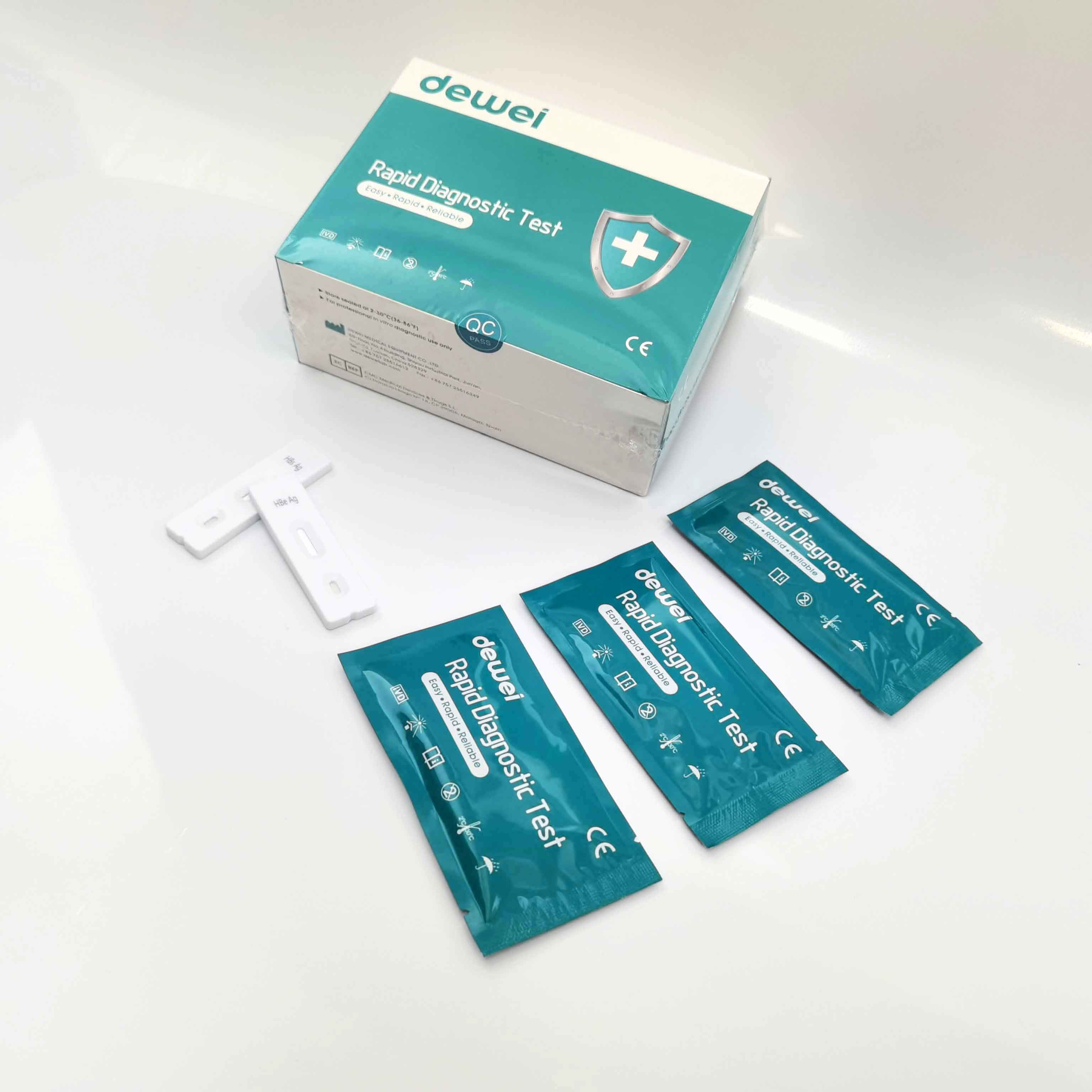
HBV One Step Hepatitis B Virus Combo Rapid Test Device 5 Parameters in 1 Cassette
INTENDED USE
The HBV One Step Hepatitis B Virus Combo Test Device (Serum/Plasma) is a rapid chromatographic immunoassay for the qualitative detection of HBsAg, HBsAb, HBeAg, HBeAb and HBcAb in serum or plasma.
INTRODUCTION
Chronic hepatitis B is a serious, debilitating illness that can cause cirrhosis of the liver, liver cancer and death. Chronic hepatitis B is the main cause of liver cancer and the tenth leading cause of death worldwide, with 400,000,000 people infected with the virus. Every year, one million people worldwide are expected to die from this infection.
Most people fight off the infection themselves, but approximately 5-10 percent of those infected with the virus become carriers, and an additional 5-10 percent of those infected each year will progress to chronic liver disease, cirrhosis and possibly liver cancer.
The HBV One Step Hepatitis B Virus Combo Test Device (Serum/Plasma) is a rapid test to qualitatively detect the presence of HBsAg, HBsAb, HBeAg, HBeAb and HBcAb in serum or plasma without the use of an instrument.1
PRINCIPLE
HBsAg and HBeAg
The HBsAg and HBeAg tests are qualitative, two-site sandwich immunoassays for the detection of HBsAg or HBeAg in serum or plasma. The membrane is pre-coated with anti-HBsAg or anti-HBeAg antibodies on the test line region of the strip. During testing, the serum or plasma specimen reacts with the particle coated with anti-HBsAg or anti-HBeAg antibodies.2 The mixture migrates upward on the membrane chromatographically by capillary action to react with anti-HBsAg or anti-HBeAg antibodies on the membrane and generate a colored line. The presence of this colored line in the test line region indicates a positive result, while its absence indicates a negative result.
HBsAb
Hepatitis B surface Antibody (HBsAb) is also known as anti-Hepatitis B surface Antigen (anti-HBs). This test is a qualitative, lateral flow immunoassay for the detection of HBsAb in serum or plasma. The membrane is pre-coated with HBsAg on the test line region of the strip. During testing, the serum or plasma specimen reacts with the particle coated with HBsAg. The mixture migrates upward on the membrane chromatographically by capillary action to react with HBsAg on the membrane and generate a colored line. The presence of this colored line in the test line region indicates a positive result, while its absence indicates a negative result.
HBeAb and HBcAb
Hepatitis B envelope Antibody (HBeAb) is also known as anti-Hepatitis B envelope Antigen (anti-HBe). Hepatitis B core Antibody (HBcAb) is also known as anti-Hepatitis B core Antigen (anti-HBc). These tests are immunoassays based on the principle of competitive binding.
During testing, the mixture migrates upward on the membrane chromatographically by capillary action. The membrane is pre-coated with HBeAg or HBcAg on the test line region of the strip. During testing, anti-HBe antibody or anti-HBc antibody, if present in the specimen, will compete with particle coated anti-HBe antibody or anti-HBc antibody for limited amount of HBeAg or HBcAg on the membrane, and no line will form in the test line region, indicating a positive result. A visible colored line will form in the test line region if there is no anti-HBe antibody or anti-HBc antibody in the specimen because all the antibody coated particles will be captured by the antigen coated in the test line region.
To serve as a procedural control, a colored line will always appear in the control line region indicating that proper volume of specimen has been added and membrane wicking has occurred.
MAIN CONTENTS
• One pouch containing a reaction test Cassette with desiccant.
• Buffer
• Disposable pipettes.
• Instructions for use.
STORAGE AND STABILITY
• Store at 2 ~ 30 º C in the sealed pouch for 18 months.
PRECAUTIONS
• For in vitro diagnostic use only.
• Do not use after expiration date.
• The test Cassette should remain in the sealed pouch until use.
• The used test Cassette should be discarded according to local regulations.
DIRECTION OF USE
Bring tests, specimens, and/or controls to room temperature (15-30°C) before use.
- Remove the test from its sealed pouch, and place it on a clean, level surface. Label the device with patient or control identification. For best results, the assay should be performed within one hour.
- Using the provided disposable pipette, transfer 3 drops of serum or plasma (approximately 75 µL) to each specimen well (S) of the device respectively, then start the timer.
Avoid trapping air bubbles in the specimen well (S), and do not add any solution to the result window.
As the test begins to work, color will migrate across the membrane.
- Wait for the colored band(s) to appear. The result should be read at 15 minutes. Do not interpret the result after 20 minutes.
INTERPRETATION


 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!