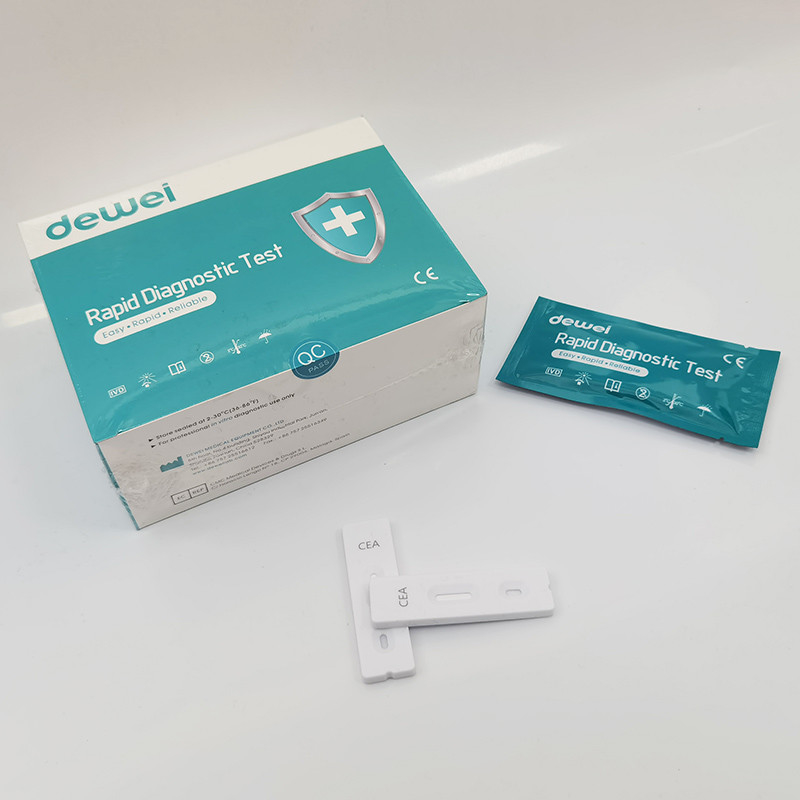
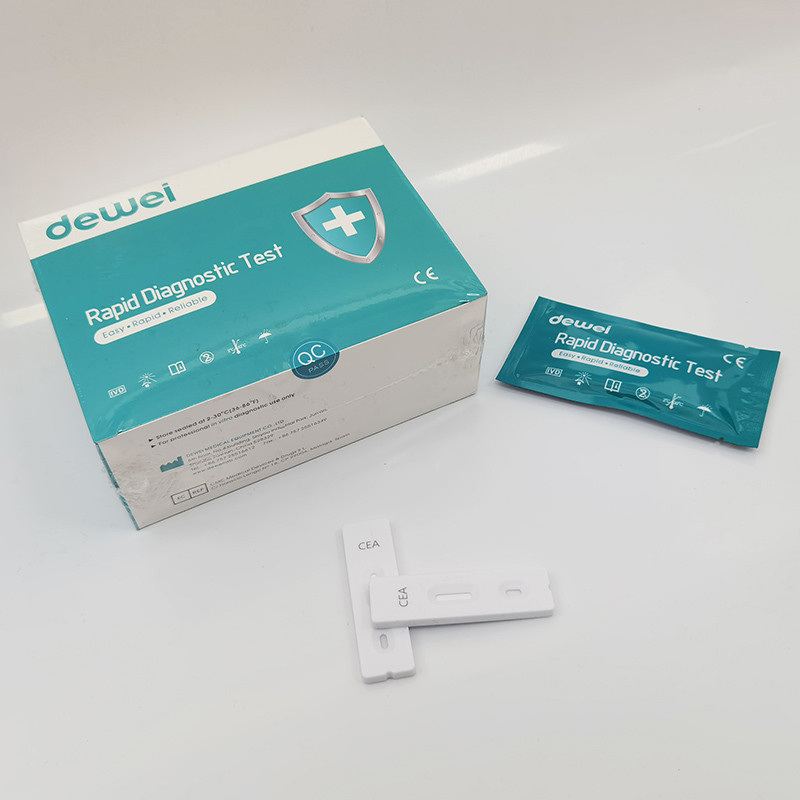
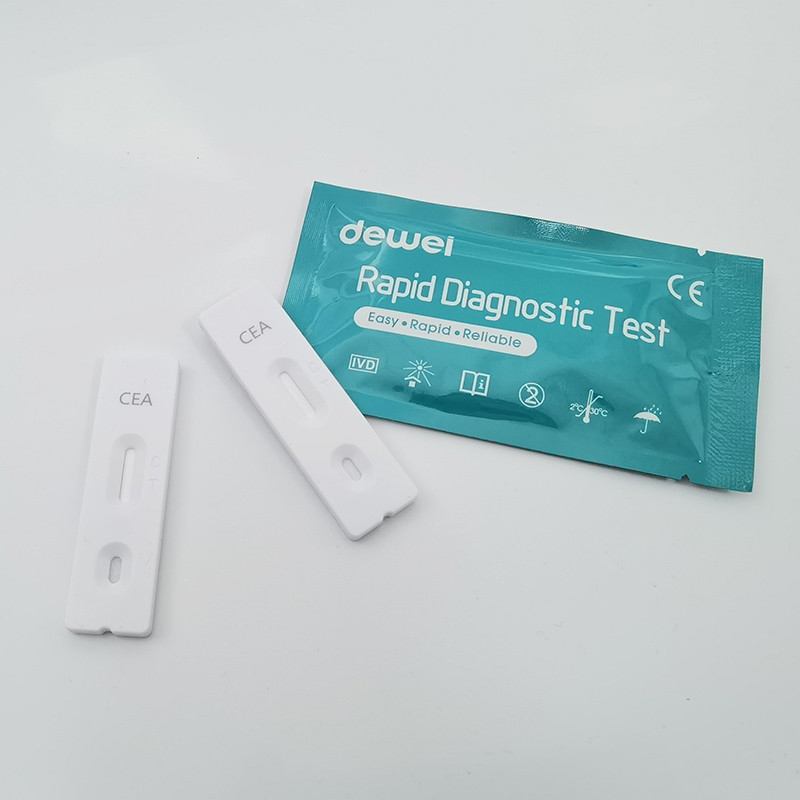
POCT Carcinoembryonic Antigen CEA Rapid Diagnostic Test Kit One Step Blood Sample
INTENDED USE
The CEA Rapid Test Kit (Whole Blood/Serum/Plasma) is a rapid visual immunoassay for the qualitative presumptive detection of human carcinoembryonic antigen (CEA) in human whole blood, serum, or plasma specimens. This kit is intended for use as an aid in the diagnosis of various cancers.
INTRODUCTION
Carcinoembryonic antigen (CEA) is a tumor-associated antigen characterized as an oncofetal glycoprotein of approximately 200,000 molecular weight with Beta electrophoretic mobility, a single protein chain of about 800 amino acids, and 50-80% carbohydrate composition. CEA was first present as a specific antigen for adenocarcinoma of the colon. More recent studies have demonstrated CEA presence in a variety of malignancies, particularly those involving ectodermal tissues of gastrointestinal or pulmonary origin. Small amounts have also been demonstrated in secretions from the colonic mucosa. Additionally, CEA-like substances have been reported in normal bile from non-icteric patients. CEA testing can have significant value in the monitoring of patients. Persistent elevation in circulating CEA following treatment is strongly indicative of occult metastic and/or residual disease. A persistent rising CEA value may be associated with progressive malignant disease and poor therapeutic response. A declining CEA value is generally indicative of a favorable prognosis and good response to treatment. Measurement of CEA has been shown to be clinically relevant in the follow-up management of patients with colorectal, breast, lung, prostatic, pancreatic, ovarian, and other carcinomas. Follow-up studies of patients with colorectal, breast and lung carcinomas suggest that the preoperative CEA level has prognostic significance.
TEST PRINCIPLE
The CEA Rapid Test Kit detects human carcinoembryonic antigen (CEA) through visual interpretation of color development on the internal strip. CEA antibodies are immobilized on the test region of the membrane. During testing, the specimen reacts with CEA antibodies conjugated to colored particles and precoated onto the sample pad of the test. The mixture then migrates through the membrane by capillary action, and interacts with reagents on the membrane. If there are sufficient CEA antigens in the specimen, a colored band will form at the test region of the membrane. The presence of this colored band indicates a positive result, while its absence indicates a negative result. The appearance of a colored band at the control region serves as a procedural control, indicating that the proper volume of specimen has been added and membrane wicking has occurred.
MAIN CONTENTS
• Rapid test Cassette with desiccant.
• Buffer
• Pipette
• Instruction Manual
STORAGE AND STABILITY
• Store at 2 ~ 30 º C in the sealed pouch for 18 months.
PRECAUTIONS
• For in vitro diagnostic use only.
• Do not use after expiration date.
• The test Cassette should remain in the sealed pouch until use.
• The used test Cassette should be discarded according to local regulations.
SAMPLE COLLECTION
1) The CEA Rapid Test Kit (Whole Blood/Serum/Plasma) is intended for use with human whole blood, serum, or plasma specimens only.
2) Only clear, non-hemolyzed specimens are recommended for use with this test. Serum or plasma should be separated as soon as possible to avoid hemolysis.
3) Perform testing immediately after specimen collection. Do not leave specimens at room temperature for prolonged periods. Serum and plasma specimens may be stored at 2-8°C for up to 3 days. For long term storage, specimens should be kept below -20°C. Whole blood collected by venipuncture should be stored at 2-8°C if the test is to be run within 2 days of collection. Do not freeze whole blood specimens. Whole blood collected by fingerstick should be tested immediately.
4) Containers containing anticoagulants such as EDTA, citrate, or heparin should be used for whole blood storage.
5) Bring specimens to room temperature prior to testing. Frozen specimens must be completely thawed and mixed well prior to testing. Avoid repeated freezing and thawing of specimens.
6) If specimens are to be shipped, pack them in compliance with all applicable regulations for transportation of etiological agents.
7) Icteric, lipemic, hemolysed, heat treated and contaminated specimens may cause erroneous results.
OPERATION
Bring tests, specimens, buffer and/or controls to room temperature (15-30°C) before use.
1) Remove the test from its sealed pouch, and place it on a clean, level surface. Label the device with patient or control identification. For best results, the assay should be performed within one hour.
2) Transfer 3 drops of whole blood/serum/plasma to the specimen well (S) of the device with the provided disposable pipette, and start the timer.
OR
Allow 3 hanging drops of fingerstick whole blood to fall into the center of the specimen well (S) of the test device, and start the timer.
Avoid trapping air bubbles in the specimen well (S), and do not add any solution to the result area.
As the test begins to work, color will migrate across the membrane.
3) If the test fails to migrate across the membrane after 1 minute, add 1 drop of buffer to the specimen well (S).
4) Wait for the colored band(s) to appear. The result should be read at 15 minutes. Do not interpret the result after 20 minutes.
INTERPRETATION
POSITIVE: Two colored bands appear on the membrane. One band appears in the control region (C) and another band appears in the test region (T).
NEGATIVE: Only one colored band appears, in the control region (C). No apparent colored band appears in the test region (T).
INVALID: Control band fails to appear. Results from any test which has not produced a control band at the specified read time must be discarded. Please review the procedure and repeat with a new test. If the problem persists, discontinue using the kit immediately and contact your local distributor.
FRQ:
| 1 |
What level of CEA indicates cancer? |
There's no magic number or threshold for a CEA test that points to cancer. Healthcare providers don't use the CEA test to screen for or diagnose cancer. In general, a CEA level of more than 2.9 ng/mL is considered abnormal but doesn't necessarily mean that cancer is present. |
| 2 |
Is CEA a good cancer marker? |
The test is often used with other tests to check if cancer treatment is working. CEA tests are not used to screen for or diagnose cancer. That's because: Cancers that often cause high CEA levels don't always cause high levels. |
| 3 |
What is CEA in relation to cancer? |
Carcinoembryonic antigen (CEA) is a protein normally found in very low levels in the blood of adults. The CEA blood level may be increased in certain types of cancer and non-cancerous (benign) conditions. A CEA test is most commonly used for colorectal cancer. |
| 4 |
What happens if CEA is high? |
A high CEA level in a person recently treated for certain cancers may mean the cancer has returned. A higher than normal level may be due to the following cancers: Breast cancer. Cancers of the reproductive and urinary tracts. |
| 5 |
Can CEA be high without cancer? |
Yes, elevations in CEA and CA 19-9 levels may occur in patients without cancer. In fact studies suggest that falsely elevated levels may occur in up to 50% of colon cancer patients post treatment leading to unnecessary anxiety and testing. |
| 6 |
Does CEA increase in cancer? |
A normal result is typically less than 2.5 nanograms per milliliter. Results might vary between labs. A higher-than-normal CEA level that increases over time might signal that your cancer has grown or has come back after treatment. But high levels of CEA do not always mean you have cancer. |
| 7 |
How to reduce CEA level? |
Eating foods high in omega-3 fatty acids: Similar to vitamin D, omega-3 fatty acids can also help suppress CEA. You can enrich your diet with this essential nutrient by leaning toward foods such as fish (mackerel, sardines, herring, salmon), seafood, and seeds, like chia and flaxseeds. |
| 8 |
What medications increase CEA? |
Actually, it has been shown that the expression of CEA can be up-regulated by pharmacological agents including,聽antineoplastic drugs (i.e. 5-fluorouracil), cytokines (i.e. interferons or interleukin-6), differentiating agents (i.e. sodium butyrate) and protein kinase inhibitors (i.e. staurosporine). |
| 9 |
What blood test shows cancer? |
Circulating tumor cell tests detect cancer cells in your blood. The cells might be in the blood if they've broken away from where they started and are spreading to other parts of the body. Circulating tumor cell tests are mostly used after a cancer diagnosis. |
| 10 |
What is the cost of a CEA test? |
The Carcinoembryonic Antigen CEA Test Price in Mumbai is ₹ 950 . We are committed to deliver accurate and quality results from the best labs in India with complete transparency regarding test cost and turnaround time. |
| 11 |
What is a good CEA level? |
In healthy, non-smoking adults, CEA is considered within normal limits at a level of<=3.0 µg/L. Smokers may have elevated CEA, and therefore it is considered within normal limits at a level of <5 µg/L. |
| 12 |
Can stress increase CEA? |
Our findings clearly indicated that immobilization stress results in enhanced serum CEA level whether the stress is acute or chronic. Consistent with our finding, there are reports suggesting that stress can increase tumor growth and tumor marker expression. |
| 13 |
What is the maximum CEA level? |
The normal range for CEA is 0 to 2.5 nanograms per milliliter of blood (ng/mL). If CEA levels remain elevated during treatment, the treatment may not have been as successful as hoped. Anything greater than 10 ng/mL suggests extensive disease, and levels greater than 20 ng/mL suggest the cancer may be spreading. |
| 14 |
What causes CEA to rise? |
Increasing CEA level during treatment may be caused by cancer progression (tumor growth or metastasis). If your cancer is progressing, your oncology team will adjust your treatment plan. CEA level may increase after starting chemotherapy for colorectal cancer, and then decrease as treatment continues. |
| 15 |
What does a CEA level of 500 mean? |
CEA levels greater than 271 ng/ml are significant for advanced tumor size, advanced tumor staging, metastasis to the contralateral neck compartment, and decreased chance of biochemical cure. CEA levels greater than 500 ng/ml are greatly associated with patient mortality. |
For detailed information, please contact Dewei person for Manual Instruction.




 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!