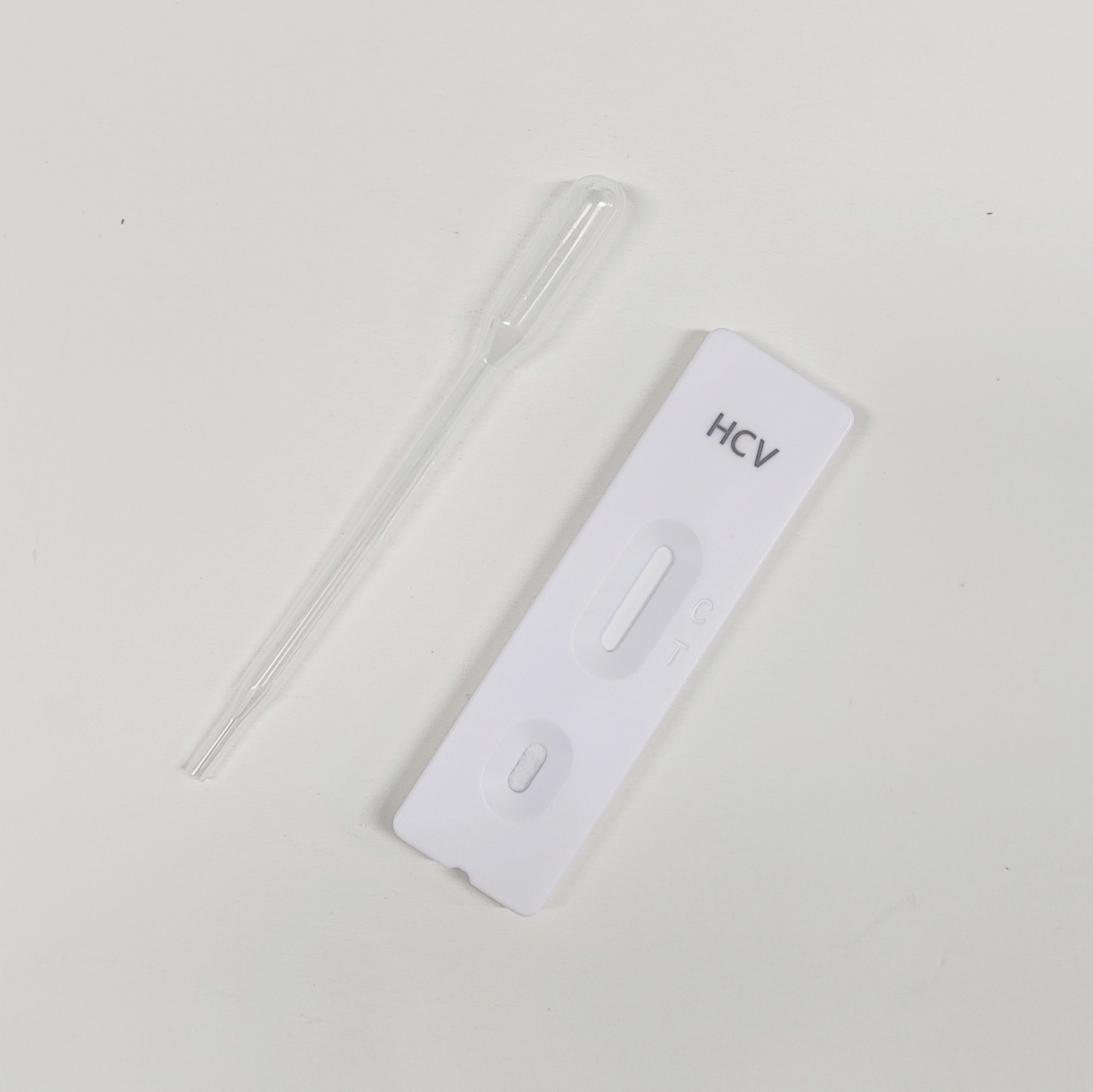
Chikungunya Virus ChikV IgG/IgM Rapid Test Kit Cassette Type Qualitative Detection
【INTENDED USE】
Chikungunya Virus (ChikV) IgG/IgM Rapid Test is a rapid immunochromatographic assay for the simultaneous detection of IgG and IgM antibodies to Chikungunya virus in human whole blood, serum or plasma. The assay is used as a screening test for Chikungunya viral infection.
For professional use only.
For in vitro diagnostic use only.
【PRINCIPLE】
ChikV rapid test utilizes the principle of Immuno-chromatography. Mouse anti-human IgM and human IgG antibodies are immobilized on the nitrocellulose membrane respectively, as two individual test lines (IgM line and IgG line) in the test window of the test device. The IgG line in the test window is closer to the sample well and followed by IgM line. As the test sample flows through the membrane within the test, the colored–chikungunya specific recombinant antigencolloidal gold conjugate complexes with specific antibodies (IgM and/or IgG) of chikungunya virus, if present in the sample. This complex moves further on the membrane to the test region where it is captured by the anti-human IgM and/or human IgG antibodies coated on the membrane leading to formation of a colored band, which indicates a positive test results. Absence of this colored band in the test window indicates a negative test result. A built-in control line will always appear in the test window when the test has performed properly, regardless of the presence or absence of anti- ChikV antibodies in the specimen.
【WARNINGS AND PRECAUTIONS】
- Immunoassay for in vitro diagnostic use only.
- Do not use after expiration date.
- The test should remain in the sealed pouch until use.
- The used test should be discarded according to local regulations.
【SPECIMEN COLLECTION】
Serum and plasma:
Test specimens as soon as possible after collecting. Store specimens at 2°C -8°C if not tested immediately. Specimens can be stored at 2°C -8°C for up to 5 days.
The plasma and serum specimens should be frozen at -20°C for longer storage.
Avoid multiple freeze-thaw cycles. Prior to testing, bring frozen specimens to room temperature slowly and mix gently. Specimens containing visible particulatematter should be clarified by centrifugation before testing.
Do not use samples demonstrating gross lipemia, gross hemolysis or turbidity in order to avoid interference on result interpretation.
Whole Blood:
Whole blood can be obtained by either fingertip puncture or venipuncture.
Whole blood specimens should be stored in refrigeration (2°C-8°C), if not tested immediately. The specimens must betested within 24 hours of collection.
【STORAGE AND STABILITY】
- The kit should be stored at 2-30°C until the expiry date printed on the sealed pouch.
- The test must remain in the sealed pouch until use.
- Keep away from direct sunlight, moisture and heat.
- Do not freeze.
- Care should be taken to protect the components of the kit from contamination. Do not use if there is evidence of microbial contamination or precipitation. Biological contamination of dispensing equipment, containers or reagents can lead to false results.
【OPERATION】
Read the instructions thoroughly before testing and bring the card pouch and specimen to room temperature.
1. Bring the specimen and test components to room temperature if refrigerated or frozen. Once thawed, mix the specimen well prior to performing the test.
2. When ready to test, open the pouch at the notch and remove the device. Place the test device on a clean, flat surface.
3. Fill the plastic dropper with the specimen. Holding the dropper vertically, dispense 10µL of serum/plasma or 20µL of whole blood into the sample well, making sure there are no air bubbles.
4. Immediately add 3 drops (about 100 µL) of sample diluent to sample well with the bottle positioned vertically.
5. Wait for colored line(s) to appear. Interpret the test results in 15 minutes. Do not read results after 20 minutes.
【INTERPRETATION OF RESULTS】
Positive :
IgG Positive: Both IgG test region (G) and control region appear color line.
IgM Positive: Both IgM test region (M) and control region appear color line.
IgG/IgM Positive: Both test region (G and M) and control region appear color line.
Negative :
Only one color line appears on the control region (C).
Invalid:
No visible line at all, or there is a visible line only in the test region and not in the control region. Repeat with a new test kit.

To learn more about our product list, please leave message to us!


 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!